- History & Evolution
- Biosynthesis & dietary uptake
- Nicotinic acid and the microbiome
- Nicotinic acid and immunity
- Nicotinic acid and cardiometabolism
- Nicotinic acid and pharmacology
- Nicotinic acid and 5P medicine
- References
History & Evolution
1867: first synthesis | 1937: defined as vitamin B3 | 1950s: discovery of lipid-lowering effect
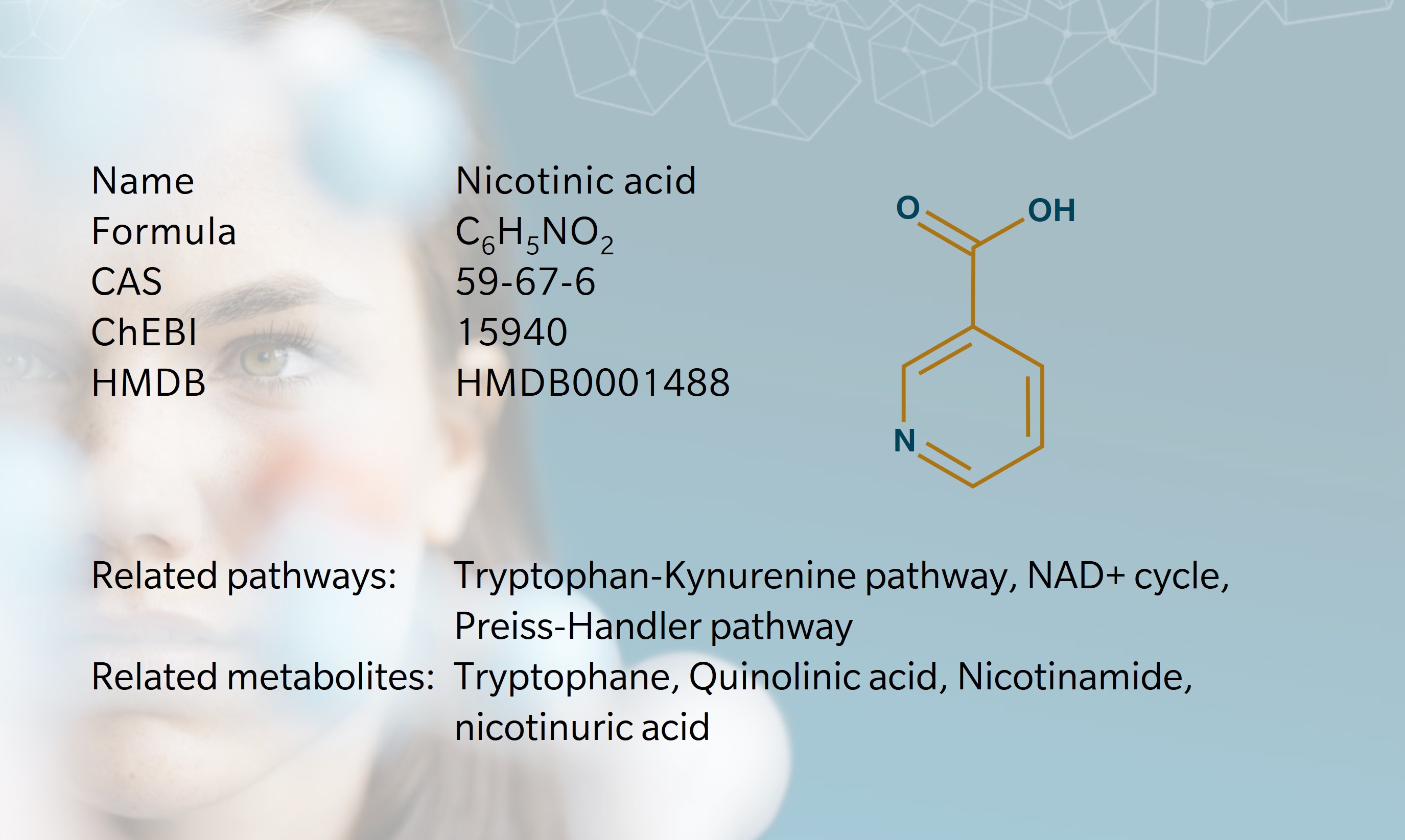
First synthesized in 1867 by Huber, nicotinic acid gained biomedical relevance in the 1930s through research on pellagra, a severe vitamin B3 or tryptophan deficiency characterized by the “3Ds”: dermatitis, diarrhea, and dementia. In 1937, a team led by Conrad Elvehjem isolated nicotinic acid from liver extract and used it to cure “black tongue”, the canine equivalent of pellagra. Soon after, nicotinic acid and nicotinamide were confirmed as pellagra curative factors in humans and established as essential components of niacin, also known as vitamin B₃ (Matthews 1938; Bieganowski et al. 2004). These findings laid the foundation for recognizing their fundamental role in cellular redox metabolism and energy homeostasis.
In the 1950s, nicotinic acid gained additional importance as a pharmacological agent when Rudolf Altschul demonstrated that gram dose supplementation lowered plasma cholesterol. It became the first effective lipid lowering drug, shown to reduce low-density lipoprotein (LDL) cholesterol, increase high-density lipoprotein (HDL) cholesterol, and promote regression of xanthomas -lipid-rich deposits that develop under the skin, well before the protective role of HDL was fully understood (Carlson 2005). Unlike nicotinamide, nicotinic acid exhibits distinct lipid modifying effects but is also associated with the niacin flush characteristic of high dose vitamin B₃ uptake, which drove the development of modified and extended release formulations to improve tolerability (Carlson 2005).
Today, nicotinic acid is recognized as both an essential form of vitamin B₃ and a historically important drug, with its role in NAD⁺ biology linking early nutritional discoveries to modern research on metabolism and disease.
Biosynthesis vs. dietary uptake
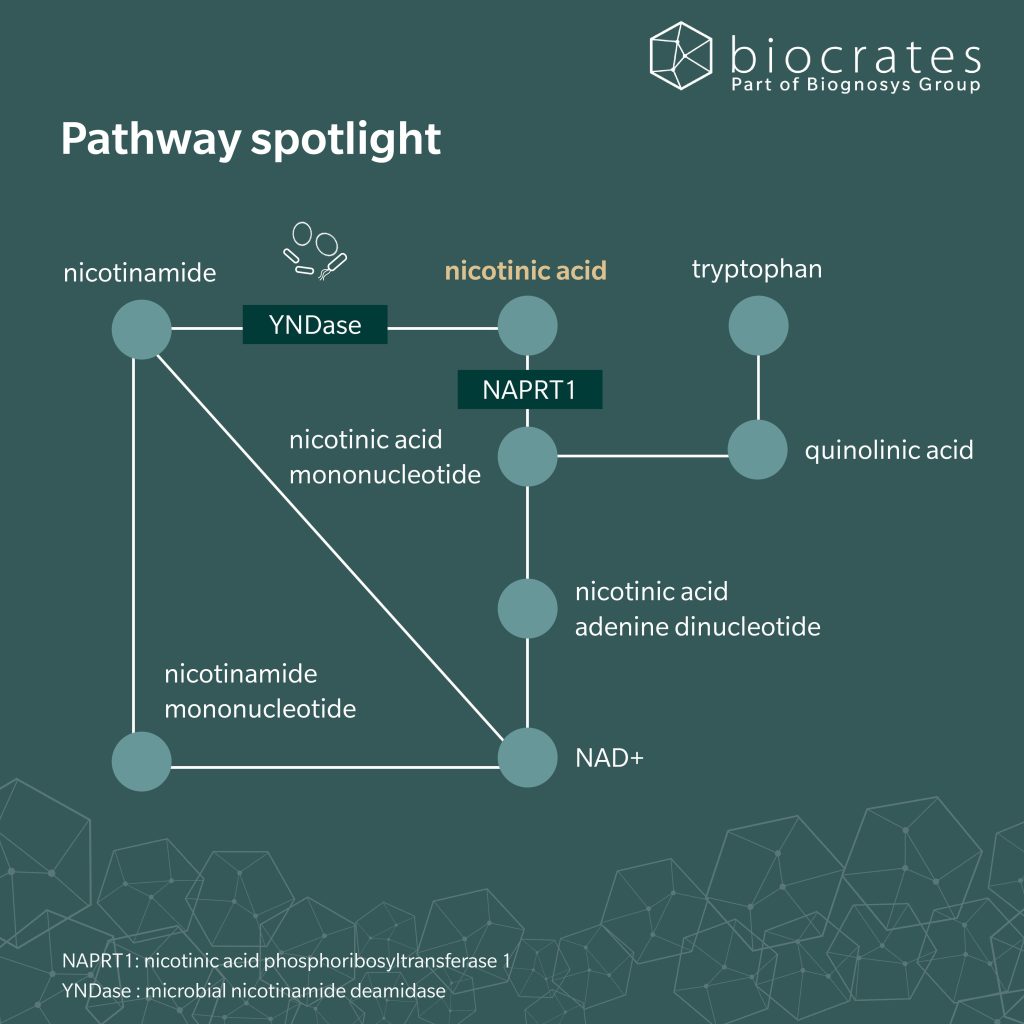
Nicotinic acid contributes to vitamin B₃ supply through dietary intake and endogenous biosynthesis, ultimately sustaining cellular pools of cofactors NAD⁺ and NADP+. In the diet, nicotinic acid is found in foods such as meat, fish and nuts (Zeman et al. 2015).
After intestinal absorption, nicotinic acid directly enters NAD⁺ biosynthesis via the Preiss–Handler pathway, where it is converted by nicotinic acid phosphoribosyltransferase 1 (NAPRT1) to nicotinic acid mononucleotide (NAMN) and subsequently incorporated into NAD⁺ and NADP⁺ (Oyama et al. 2024).
In addition, NAD⁺ can be synthesized from tryptophan via the kynurenine pathway. Tryptophan is metabolized through multiple enzymatic steps to quinolinic acid, which is then converted to NAMN and enters NAD⁺ biosynthesis at the same metabolic node as the Preiss–Handler pathway (Fukuwatari et al. 2013). This de novo route is considered metabolically inefficient in humans (Goldsmith et al. 1961) and nutritionally demanding, requiring several micronutrient cofactors (McCormick 1989). Dietary processing strongly affects nicotinic acid bioavailability, as illustrated by maize based diets lacking alkaline treatment, in which niacin remains poorly absorbable (Carpenter 1983).
Because NAD⁺ acts as a central redox cofactor, including its role in the tricarboxylic acid cycle, adequate nicotinic acid availability is essential for mitochondrial respiration and energy metabolism (Oyama et al. 2024).
Excess nicotinic acid that is not incorporated into NAD⁺ or NADP⁺ pools undergoes hepatic detoxification, primarily through methylation to N¹ methylnicotinamide, followed by oxidation and conjugation to form nicotinuric acid, which is ultimately excreted in the urine (Neuvonen et al. 1991).
Nicotinic acid and the microbiome
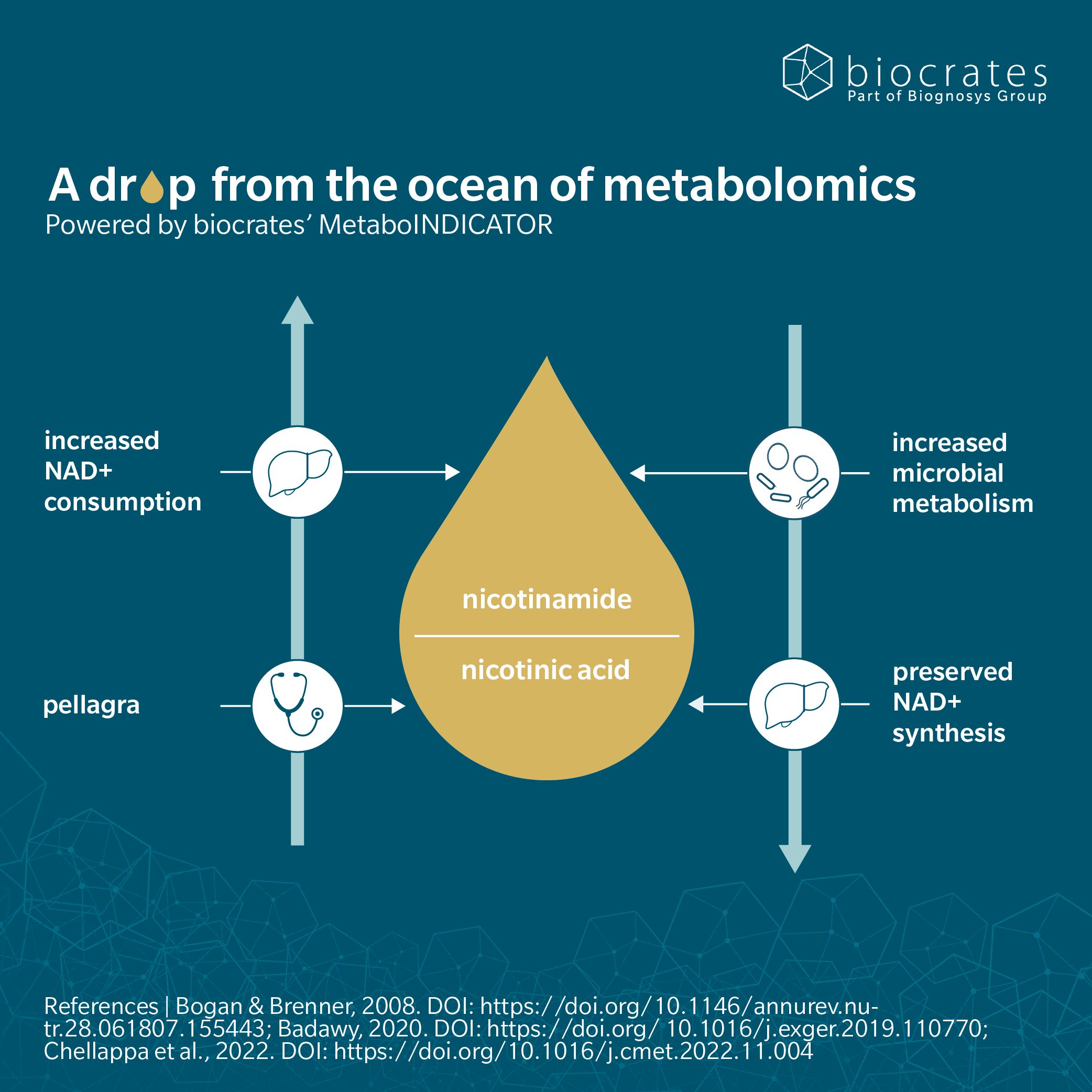
The gut microbiome contributes to host nicotinic acid and NAD⁺ metabolism, forming a bidirectional metabolic exchange between host tissues and gut bacteria. While dietary nicotinic acid is largely absorbed in the upper intestine, circulating host derived nicotinamide, which is continuously generated through NAD⁺ consuming reactions, can reach the gut lumen, where it is converted by the microbial enzyme nicotinamide deamidase (YNDase) into nicotinic acid. Microbiome derived nicotinic acid can then re enter host metabolism and support Preiss–Handler–dependent NAD⁺ biosynthesis, thereby helping to maintain systemic NAD⁺ homeostasis even under conditions of low dietary intake (Chellappa et al. 2022).
Beyond systemic metabolism, microbiome derived nicotinic acid exerts local effects in the intestine. Recent work in mice demonstrated that bacterially produced nicotinic acid shapes colonic epithelial identity and regionalization, partly through nuclear receptor signaling, and protects against epithelial injury in preclinical models (Rispal et al. 2026). These findings link microbial nicotinic acid production to intestinal barrier integrity and susceptibility to inflammatory damage.
Nicotinic acid and immunity
Nicotinic acid directly modulates immune and inflammatory responses primarily through activation of the G protein–coupled receptor GPR109A (HCAR2), which is expressed on macrophages, epithelial cells, and other immune relevant cell types (Kostylina et al. 2008; Lukasova et al. 2011). In macrophages, cell culture and animal models demonstrated that nicotinic acid–GPR109A signaling promotes an anti inflammatory phenotype with reduced release of NF κB–dependent mediators such as TNF α and IL 6 (Digby et al. 2012; Si et al. 2014). These effects contribute to the anti atherosclerotic properties of nicotinic acid by simultaneously modulating macrophage inflammation and foam cell formation (Lukasova et al. 2011; Si et al. 2014). In contrast, GPR109A activation in cutaneous immune cells mediates prostaglandin release and underlies the characteristic niacin flush observed in humans (Morrow et al. 1989; Benyó et al. 2005).
Beyond the periphery, neuronal and glial cell culture studies have shown that nicotinic acid suppresses NF κB–driven inflammatory signaling, suggesting a role in regulating neuroinflammation (Wuerch et al. 2023). In addition, silk based biomaterial scaffolds incorporating nicotinic acid have emerged as localized anti inflammatory delivery systems, where controlled release reduces inflammatory responses and improves tissue compatibility in preclinical models (Zakeri Siavashani et al. 2019).
Nicotinic acid and cardiometabolism
Nicotinic acid has long been recognized as a potent modulator of dyslipidemia, particularly in the context of metabolic syndrome and type 2 diabetes (Carlson 2005; Keenan 2024). At pharmacological doses, nicotinic acid lowers very low-density lipoprotein (VLDL), LDL, triglycerides, and lipoprotein(a), the latter being a lipoprotein fraction that is largely refractory to pharmacological and lifestyle based interventions (Capuzzi et al. 1998; Carlson 2005; Zeman et al. 2015). In parallel, nicotinic acid produces the most pronounced increase in HDL cholesterol among available lipid modifying agents, largely by reducing hepatic apolipoprotein A-1 catabolism and enhancing macrophage cholesterol efflux via ATP-binding cassette transporter A1 (ABCA1) (Carlson 2005).
These lipid effects arise primarily from inhibition of adipose tissue lipolysis, resulting in reduced plasma free fatty acid availability and secondary reductions in hepatic triglyceride synthesis and VLDL secretion (Carlson 2005). Early monotherapy trials, including the Coronary Drug Project, demonstrated reductions in cardiovascular events, stroke risk, and mortality (Keenan 2024). In contrast, large contemporary outcome trials (AIM HIGH and HPS2 THRIVE) failed to show incremental benefit when nicotinic acid was added to intensive statin therapy, despite robust lipid improvements (Zeman et al. 2015).
These results prompted a reevaluation of nicotinic acid in cardiovascular prevention but have also been widely criticized. Major limitations include conditions under maximally optimized statin therapy, use of sustained release formulations, fasting or bedtime dosing, potential GPR109A desensitization and metabolic counter regulation, limited treatment duration, and population specific safety signals, particularly in HPS2 THRIVE (Zeman et al. 2015; Keenan 2024). Together, these factors suggest that the neutral outcomes of contemporary trials may reflect context dependent inefficacy, rather than a lack of intrinsic cardiometabolic potential.
Nicotinic acid and pharmacology
Pharmacologically, nicotinic acid acts through both receptor mediated signaling and integration into NAD⁺ dependent metabolic pathways. Activation of GPR109A mediates acute anti lipolytic and anti inflammatory effects but also causes the characteristic cutaneous flushing, driven by prostaglandin D₂ and E₂ release from epidermal Langerhans cells (Zeman et al. 2015). Consequently, formulation and dosing are critical: immediate release nicotinic acid is effective but poorly tolerated, whereas sustained release formulations reduce flushing at the cost of increased hepatotoxicity. Extended release preparations were developed to balance efficacy and safety and have demonstrated long term lipid efficacy as monotherapy in humans under appropriate monitoring (Capuzzi et al. 1998).
Beyond receptor signaling, pharmacological nicotinic acid rapidly feeds into NAD⁺ metabolism, activating peroxisome proliferator-activated receptors (PPAR)α and PPARδ dependent programs that regulate fatty acid oxidation and carnitine homeostasis in animal and mechanistic cell based studies (Couturier et al. 2014). Human tracer studies further show that intermittent, postprandial dosing favorably remodels lipid trafficking, markedly reducing hepatic and cardiac lipid exposure without sustained metabolic escape (Montastier et al. 2025). Together, these findings define nicotinic acid pharmacology as dose , formulation , and context dependent, extending beyond classical lipid endpoints.
Nicotinic acid and 5P medicine
Therapeutic use of nicotinic acid exemplifies a context dependent metabolic intervention whose effects depend on dose, formulation, timing, and individual metabolic state, spanning cardiometabolic, immune, and neurological pathways. Through its role as a precursor of NAD⁺, nicotinic acid supports cellular energy metabolism, mitochondrial function, and redox homeostasis, processes that decline with aging and contribute to reduced metabolic resilience, making nicotinic acid a potential aging relevant metabolic supplement, particularly for supporting stress adaptation rather than lifespan extension per se (Yang et al. 2019; Oyama et al. 2024).
From a preventive cardiometabolic perspective, clinical studies demonstrate improvements in dyslipidemia and, with short acting postprandial dosing, enhanced adipose tissue fatty acid trapping with reduced hepatic and cardiac lipid exposure (Montastier et al. 2025). The precision and personalization dimensions of 5P medicine are underscored by large inter individual differences in lipid response (Zeman et al. 2015), flushing susceptibility (Benyó et al. 2005), glucose handling (Zeman et al. 2015), and inflammatory tone, while the immune dimension reflects context dependent modulation of macrophage activity (Lukasova et al. 2011; Digby et al. 2012). Emerging evidence further suggests nicotinic acid as a supportive strategy in neurological disorders, including Alzheimer’s disease, Parkinson’s disease, glioblastoma, and amyotrophic lateral sclerosis. These interests are largely driven by nicotinic acid’s role as a precursor of NAD⁺ (Wuerch et al. 2023). Together, these findings position nicotinic acid as a context dependent metabolic modulator whose clinical value emerges from precision in dose, timing, and patient stratification, rather than uniform application.
References
Benyó, Z. et al.: GPR109A (PUMA-G/HM74A) mediates nicotinic acid-induced flushing (2005) The Journal of clinical investigation | https://doi.org/10.1172/JCI23626.
Bieganowski, P. et al.: Discoveries of nicotinamide riboside as a nutrient and conserved NRK genes establish a Preiss-Handler independent route to NAD+ in fungi and humans (2004) Cell | https://doi.org/10.1016/S0092-8674(04)00416-7.
Capuzzi, D.M. et al.: Efficacy and safety of an extended-release niacin (Niaspan): a long-term study (1998) The American journal of cardiology | https://doi.org/10.1016/S0002-9149(98)00731-0.
Carlson, L.A.: Nicotinic acid: the broad-spectrum lipid drug. A 50th anniversary review (2005) Journal of internal medicine | https://doi.org/10.1111/j.1365-2796.2005.01528.x.
Carpenter, K.J.: The relationship of pellagra to corn and the low availability of niacin in cereals (1983) Experientia. Supplementum | https://doi.org/10.1007/978-3-0348-6540-1_12.
Chellappa, K. et al.: NAD precursors cycle between host tissues and the gut microbiome (2022) Cell metabolism | https://doi.org/10.1016/j.cmet.2022.11.004.
Couturier, A. et al.: Pharmacological doses of niacin stimulate the expression of genes involved in carnitine uptake and biosynthesis and improve the carnitine status of obese Zucker rats (2014) BMC pharmacology & toxicology | https://doi.org/10.1186/2050-6511-15-37.
Digby, J.E. et al.: Anti-inflammatory effects of nicotinic acid in human monocytes are mediated by GPR109A dependent mechanisms (2012) Arteriosclerosis, thrombosis, and vascular biology | https://doi.org/10.1161/ATVBAHA.111.241836.
Fukuwatari, T. et al.: Nutritional aspect of tryptophan metabolism (2013) International journal of tryptophan research : IJTR | https://doi.org/10.4137/IJTR.S11588.
Goldsmith, G.A. et al.: Efficiency of Tryptophan as a Niacin Precursor in Man (1961) The Journal of Nutrition | https://doi.org/10.1093/jn/73.2.172.
Keenan, J.: The Niacin Rebirth: Revisiting the Potential of Nicotinic Acid Therapy for Cardiovascular Disease and Niacin Supplementation for Healthy Aging (2024) Medical Research Archives | https://doi.org/10.18103/mra.v12i7.5521.
Kostylina, G. et al.: Neutrophil apoptosis mediated by nicotinic acid receptors (GPR109A) (2008) Cell death and differentiation | https://doi.org/10.1038/sj.cdd.4402238.
Lukasova, M. et al.: Nicotinic acid inhibits progression of atherosclerosis in mice through its receptor GPR109A expressed by immune cells (2011) The Journal of clinical investigation | https://doi.org/10.1172/JCI41651.
Matthews, R.S.: PELLAGRA AND NICOTINIC ACID (1938) Journal of the American Medical Association | https://doi.org/10.1001/jama.1938.02790390004002.
McCormick, D.B.: Two interconnected B vitamins: riboflavin and pyridoxine (1989) Physiological reviews | https://doi.org/10.1152/physrev.1989.69.4.1170.
Montastier, É. et al.: Nicotinic acid increases adipose tissue dietary fatty acid trapping and reduces postprandial hepatic and cardiac fatty acid uptake in prediabetes (2025) European journal of pharmacology | https://doi.org/10.1016/j.ejphar.2025.177563.
Morrow, J.D. et al.: Release of markedly increased quantities of prostaglandin D2 in vivo in humans following the administration of nicotinic acid (1989) Prostaglandins | https://doi.org/10.1016/0090-6980(89)90088-9.
Neuvonen, P.J. et al.: The bioavailability of sustained release nicotinic acid formulations (1991) British journal of clinical pharmacology | https://doi.org/10.1111/j.1365‑2125.1991.tb03933.x.
Oyama, T. et al.: Supplementation of Nicotinic Acid and Its Derivatives Up-Regulates Cellular NAD+ Level Rather than Nicotinamide Derivatives in Cultured Normal Human Epidermal Keratinocytes (2024) Life (Basel, Switzerland) | https://doi.org/10.3390/life14030413.
Rispal, J. et al.: Microbiome-produced nicotinic acid controls colon regional identity and injury susceptibility (2026) Cell | https://doi.org/10.1016/j.cell.2026.02.007.
Si, Y. et al.: Niacin inhibits vascular inflammation via downregulating nuclear transcription factor-κB signaling pathway (2014) Mediators of inflammation | https://doi.org/10.1155/2014/263786.
Wuerch, E. et al.: The Promise of Niacin in Neurology (2023) Neurotherapeutics : the journal of the American Society for Experimental NeuroTherapeutics | https://doi.org/10.1007/s13311-023-01376-2.
Yang, N.-C. et al.: The Lifespan Extension Ability of Nicotinic Acid Depends on Whether the Intracellular NAD+ Level Is Lower than the Sirtuin-Saturating Concentrations (2019) International journal of molecular sciences | https://doi.org/10.3390/ijms21010142.
Zakeri Siavashani, A. et al.: Silk based scaffolds with immunomodulatory capacity: anti-inflammatory effects of nicotinic acid (2019) Biomaterials science | https://doi.org/10.1039/C9BM00814D.
Zeman, M. et al.: Niacin in the Treatment of Hyperlipidemias in Light of New Clinical Trials: Has Niacin Lost its Place? (2015) Medical science monitor : international medical journal of experimental and clinical research | https://doi.org/10.12659/MSM.893619.