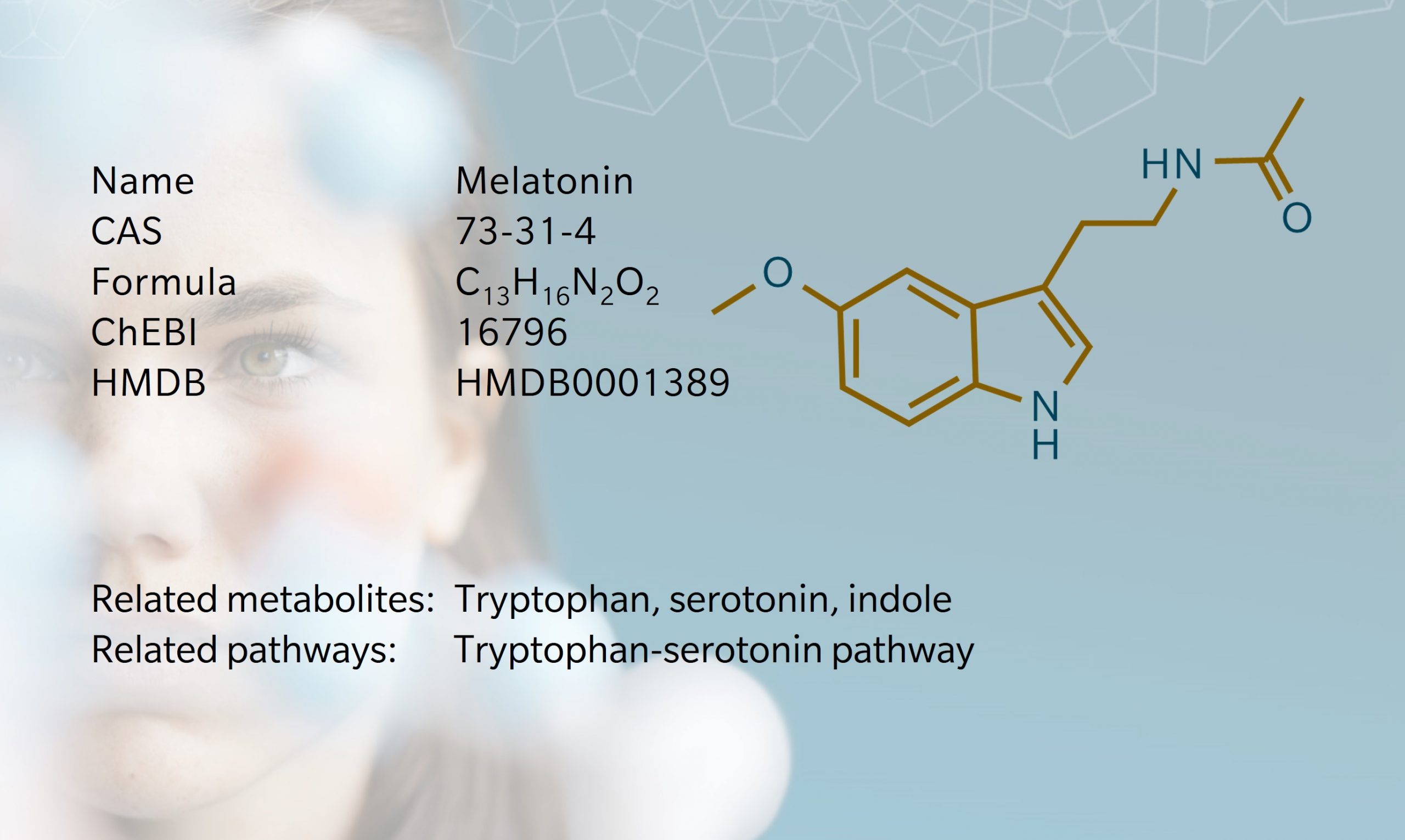
History and evolution
Biosynthesis vs. dietary uptake
Melatonin, sleep and circadian rhythm
Melatonin and the microbiom
Melatonin and neurology
Melatonin and cancer
History and evolution
1958: discovery (Lerner et al. 1958) | 1961: identified in mammalian pineal gland (Prop et al.1961) | 1993: identified as antioxidant (Tan et al. 1993)
Melatonin is an ancient molecule found in all living organisms from bacteria to humans, with an evolution that can be traced back 2.5 billion years (Tan et al. 1993). It was first discovered by Aaron Lerner and colleagues in 1958, in bovine pineal extract (Lerner et al. 1958). Melatonin gets its name from earlier work by McCord and Allen, which showed that bovine pineal extracts could trigger melanin aggregation and induce skin lightening in frogs (Masters et al. 2014). Initially, researchers thought the effect on pigmentation might make melatonin a potential treatment for vitiligo, and while that hypothesis was ultimately disproved, the investigation led to the discovery of melatonin’s role in promoting sleep (Masters et al. 2014).
Melatonin is an endogenous hormone best known for its role in regulating circadian rhythms, and for that reason is sometimes referred to as the ‘sleep hormone’ or ‘hormone of darkness’. Besides its sleep-promoting properties, melatonin has a wide range of other physiological functions and effects. In the 1990s, it was found to act as a free radical scavenger in mitochondria (Tan et al. 1993). It protects against oxidative stress, regulates energy metabolism and modulates immune processes (Alghamdi 2018). Melatonin has also been found to have anti-cancer properties and is clinically relevant to gastrointestinal disorders and neurological conditions (Alghamdi 2018).
Biosynthesis vs. dietary uptake
Melatonin is derived from the essential amino acid tryptophan. In animals, tryptophan is converted to 5-hydroxytryptophan and decarboxylated to form serotonin (Masters et al. 2014). Serotonin is catalyzed by arylalkylamine N-acetyltransferase (AANAT) to form N-acetylserotonin (NAS). Then, the enzyme hydroxyl-indole-O-methyltransferase (HIOMT) catalyzes NAS to form melatonin. AANAT is the rate-limiting enzyme in the tryptophan-serotonin-melatonin pathway, along with serotonin availability (Chowdhury et al. 2008).
Melatonin synthesis occurs primarily in the pineal gland and retina, but is also produced in other parts of the brain and other organs, including the skin, gastrointestinal (GI) tract and mitochondria (Arendt et al. 2022, Tan et al. 2013). On a recent episode of The Metabolomist, Jennifer Kirwan, Head of the Metabolomics Platform at the Berlin Institute of Health, discusses why melatonin is so intriguing:
“One of the interesting things is how much is actually extra-pineal melatonin. The retina produces some melatonin. So does the gut, the reproductive system, and some immune cells, in particular macrophages and mast cells… As we explore the tryptophan pathway and the gut microbiome further, we’re going to find that melatonin starts popping up in some very interesting metabolic processes.”
While most melatonin is synthesized endogenously, it can also be taken up directly from food, and serum levels of melatonin have been found to increase following consumption of foods that contain melatonin (Meng et al. 2017). Eggs, fish, nuts and seeds, mushrooms and legumes are all good dietary sources of melatonin.
Melatonin synthesis is heavily influenced by environmental factors, with different effects at different sites. Interestingly, while changes in light influence pineal melatonin synthesis, dietary consumption of tryptophan appears to be the rate-limiting factor for melatonin synthesis in the GI tract (Huether 1993).
As an indole alkaloid and amphiphilic molecule, melatonin passes through lipid membranes relatively easily (Tan et al. 2013). Melatonin receptors belong to the transmembrane G-protein coupled receptors. These signaling pathways are cell- and tissue-dependent, with diverse regulatory and protective effects (Ferlazzo et al. 2020).
Melatonin, sleep and circadian rhythm
Melatonin synthesis is photosensitive and diurnal, with concentrations rising as light fades, peaking during the night (or dark periods), and falling again when light returns (Vassey et al. 2021). This cycle is regulated by the body’s circadian ‘clock’, the suprachiasmatic nucleus (SCN), located in the hypothalamus. The SCN sends behavioral, hormonal and neuronal signals to peripheral circadian systems, such as the liver, stomach and adrenal system, to maintain energy homeostasis and regulate hunger, digestion and insulin release (Kumar Jha et al. 2015).
There is a clear link between the SCN, sleep and metabolism. Disruption to sleep patterns can affect melatonin production and is associated with cardiovascular disease, cognitive impairment, immune dysfunction, diabetes and metabolic syndrome (Vassey et al. 2021). Research shows that shift workers have lower levels of circulating melatonin, which correlates with a higher risk of cancer and other diseases (Wang et al. 2015).
Metabolomics is helping to elucidate the underlying mechanisms. For example, chronobiology researchers led by Professor Debra Skene at the University of Surrey have used targeted metabolomics to measure metabolite profiles in sleep-deprived phenotypes. Their findings demonstrate that sleep deprivation reduces the amplitude of daily rhythms in many metabolites, including melatonin (Davies et al. 2014). The research shows that sleep deprivation activates the melatonin synthesis pathway in both men and women, though there are significant differences in male and female metabolomic profiles: in men, most metabolites increase during sleep deprivation, whereas in women, most metabolites were found to decrease (Davies et al. 2014, Honma et al. 2019).
Because of melatonin’s phasic profile, samples must be collected at defined times, but can include salivary, serum or urinary samples (Vassey et al. 2021). Researchers looking to study melatonin should also bear in mind that samples will be light-sensitive.
For a fascinating discussion of the use of metabolomics in the study of sleep and metabolism, see this lecture by Professor Debra Skene, published by the Technical University of Munich: https://youtu.be/cC6o4lh8GKI?si=KuF7rm9p95gWrojY
Melatonin and the microbiome
The gut microbiome is an abundant extra-pineal source of melatonin, and there is more than 400 times more melatonin in the GI tract than in the pineal gland (Iesanu et al. 2022, Chowdhury et al. 2008). Melatonin appears to have a bidirectional relationship with gut microbiota, with a positive effect on dysbiosis-related conditions, such as inflammatory bowel disease (IBD), colitis, obesity and neuropsychiatric disorders (Iesanu et al. 2022). There’s growing evidence of melatonin’s potential as a therapeutic target in these conditions.
For example, administration of a probiotic in patients with irritable bowel syndrome (IBS) showed a correlation between improved symptoms and morning levels of melatonin (Wong et al. 2015). In an animal model, melatonin treatment was found to alleviate a reduction in melatonin concentration in stress-related intestinal dysbiosis (Sook Park et al. 2020).
However, the research remains inconclusive: some findings suggest that melatonin may exacerbate the inflammatory response in IBD, depending on the gut microbiota populations harbored by the host (Da Silva et al. 2023).
Melatonin and neurology
Melatonin is thought to be involved in the pathophysiology of several neurological conditions. Traumatic brain injuries can trigger melatonin release (Seifman et al. 2008), and melatonin injections have been shown to have a protective effect in response to brain injuries and ischemic injury (Wu et al. 2017).
Several studies have found that melatonin levels are lower in people with Alzheimer’s disease (AD) compared to healthy controls, so melatonin may be a useful biomarker for AD progression (Ohashi et al. 1999). Melatonin’s antioxidant and anti-inflammatory effects may contribute to an anti-amyloidogenic effect in AD, which could prevent cognitive impairment (Alghamdi 2018).
Melatonin is also clinically relevant in Parkinson’s disease (PD). Patients with PD commonly experience sleep disorders and depression, associated with misalignment of their circadian rhythm. Clinical studies show that bright light therapy, which antagonizes melatonin receptors, can restore circadian rhythm and improve sleep quality, depressive symptoms and motor function in PD patients (Rutten et al. 2012).
Melatonin (and its analogue, agomelatine), has also demonstrated a positive therapeutic effect in the treatment of depression, by regulating circadian rhythm and mood, suppressing neuroinflammation, and promoting antioxidant processes (Kholghi et al. 2022).
Melatonin and cancer
Melatonin’s anti-cancer effects have been confirmed in multiple in vivo and in vitro studies, for multiple types of cancer (Wang et al. 2022), including colorectal, breast, gastric, prostate, ovarian, lung and oral cancers (Talib et al. 2021). Melatonin has been found to induce apoptosis, reduce cell proliferation, inhibit tumor growth, and mediate the unpleasant side effects of chemotherapy and radiotherapy such as depression and poor sleep quality (Talib et al. 2021).
The main benefits occur when melatonin is taken as a supplement to augment conventional anti-cancer treatments. For example, in patients with metastatic breast cancer, those treated with tamoxifen and melatonin showed a stronger response than those treated with tamoxifen alone (Lissoni et al. 1995).
However, while these findings are encouraging, there are some conflicting results which need further investigation (Wang et al. 2022).
References
Alghamdi: The neuroprotective role of melatonin in neurological disorders. (2018) J Neurosci Res. | https://doi.org/10.1002/jnr.24220
Arendt et al.: Physiology of the Pineal Gland and Melatonin. (2022) In Feingold K. et al., Endotext [Internet] . South Dartmouth, MA: MDText.com | https://pubmed.ncbi.nlm.nih.gov/31841296/
Chowdhury et al.: Melatonin: Fifty Years of Scientific Journey from the Discovery in Bovine Pineal Gland to Delineation of Functions in Human. (2008) Indian Journal of Biochemistry and Biophysics | https://pubmed.ncbi.nlm.nih.gov/19069840/
Da Silva et al.: (2023). The Microbiota-Dependent Worsening Effects of Melatonin on Gut Inflammation. (2023) Microorganisms | https://doi.org/10.3390/microorganisms11020460
Davies et al.: Effect of sleep deprivation on the human metabolome. (2014) PNAS | https://doi.org/10.1073/pnas.140266311
Ferlazzo et al.: Is Melatonin the Cornucopia of the 21st Century? (2020) Antioxidants | https://doi.org/10.3390/antiox9111088
Honma et al.: Effect of acute total sleep deprivation on plasma melatonin, cortisol and metabolite rhythms in females. (2019) European Journal of Neuroscience | https://doi.org/10.1111/ejn.14411
Huether: The contribution of extrapineal sites of melatonin synthesis to circulating melatonin levels in higher vertebrates. (1993) Experientia | https://doi.org/10.1007/BF01923948
Iesanu et al.: Melatonin–Microbiome Two-Sided Interaction in Dysbiosis-Associated Conditions. (2022) Antioxidants (Basel) | https://doi.org/10.3390/antiox11112244
Kholghi et al.: Night shift hormone: How does melatonin affect depression? Physiology & Behavior (2022) | https://doi.org/10.1016/j.physbeh.2022.113835
Kumar Jha et al.: Circadian rhythms in glucose and lipid metabolism in nocturnal and diurnal mammals. (2015) Molecular and Cellular Endocrinology | https://doi.org/10.1016/j.mce.2015.01.024
Lerner et al.: ISOLATION OF MELATONIN, THE PINEAL GLAND FACTOR THAT LIGHTENS MELANOCYTES. (1958) J. Am. Chem. Soc. | https://doi.org/10.1021/ja01543a060
Lissoni et al.: Modulation of cancer endocrine therapy by melatonin: a phase II study of tamoxifen plus melatonin in metastatic breast cancer patients progressing under tamoxifen alone. (1995) Br J Cancer | https://doi.org/10.1038/bjc.1995.164
Masters et al.: (2014). Melatonin, the Hormone of Darkness: From Sleep Promotion to Ebola Treatment. (2014) Brain Disord Ther. | https://doi.org/10.4172/2168-975X.1000151
Meng et al.: Dietary Sources and Bioactivities of Melatonin. (2017) Nutrients | https://doi.org/10.3390/nu9040367
Ohashi et al.: Daily rhythm of serum melatonin levels and effect of light exposure in patients with dementia of the Alzheimer’s type. (1999) Biol Psychiatry | https://doi.org/10.1016/s0006-3223(98)00255-8
Prop et al.: Demonstration of some compounds present in the pineal organ of the albino rat by hystochemical methods and paper chromatography. (1961) | https://doi.org/10.1159/000141742
Rutten et al. (2012). Bright Light Therapy in Parkinson’s Disease: An Overview of the Background and Evidence. (2012) Parkinson’s Disease | https://doi.org/10.1155/2012/767105
Seifman et al.: Endogenous Melatonin Increases in Cerebrospinal Fluid of Patients after Severe Traumatic Brain Injury and Correlates with Oxidative Stress and Metabolic Disarray. (2008) Journal of Cerebral Blood Flow & Metabolism | https://doi.org/10.1038/sj.jcbfm.96006
Sook Park et al.: Melatonin in the colon modulates intestinal microbiota in response to stress and sleep deprivation. (2020) Intest Res. | https://doi.org/10.5217/ir.2019.00093
Talib et al.: Melatonin in Cancer Treatment: Current Knowledge and Future Opportunities. (2021) Molecules | https://doi.org/10.3390/molecules26092506
Tan et al.: Melatonin a potent endogenous hydroxyl radical scavenger. (1993) Endocrine Journal
Tan et al.: Mitochondria and chloroplasts as the original sites of melatonin synthesis: a hypothesis related to melatonin’s primary function and evolution in eukaryotes.(2013) Journal of Pineal Research | https://doi.org/10.1111/jpi.12026
Vassey et al.: Circadian Rhythm Dysregulation and Restoration: The Role of Melatonin. (2021) Nutrients | https://doi.org/10.3390/nu13103480
Wang et al.: Use of Melatonin in Cancer Treatment: Where Are We? (2022) Int. J. Mol. | https://doi.org/10.3390/ijms23073779
Wang et al.: Night-shift work, sleep duration, daytime napping, and breast cancer risk. (2015) Sleep Med. | https://doi.org/10.1016/j.sleep.2014.11.017
Wong et al.: Melatonin Regulation as a Possible Mechanism for Probiotic (VSL#3) in Irritable Bowel Syndrome: A Randomized Double-Blinded Placebo Study. (2015) Dig. Dis. Sci.| https://doi.org/10.1007/s10620-014-3299-8
Wu et al.: Neuroprotective Mechanisms of Melatonin in Hemorrhagic Stroke. (2017) Cellular and Molecular Neurobiology | https://doi.org/10.1007/s10571-017-0461-9