- History & Evolution
- Biosynthesis & dietary uptake
- Dimethylglycine and cardiology
- Dimethylglycine and diabetes
- Dimethylglycine and neurology
- Dimethylglycine and immunity
- Dimethylglycine and oncology
- Dimethylglycine and 5P medicine
- References
History & Evolution
1943: discovery | 1950-1960: description as endurance enhancer | 1974: marketed as supplement
Dimethylglycine (DMG) was first described in 1943 (Cupp et al. 2003). It gained attention in the 1950s-1960s through studies conducted in the Soviet Union claiming improved oxygen use, reduced fatigue, and enhanced physical performance (Wolfsegger et al. 2021). This led to its widespread use among Russian athletes and cosmonauts. During this period, DMG was often incorrectly marketed as vitamin B15 or pangamic acid, in which it was sometimes included (Cupp et al. 2003).
DMG entered the U.S. supplement market in 1974, and similar performance claims appeared in American reports in 1975 (Wolfsegger et al. 2021). In 1982, the Food and Drug Administration (FDA) banned pangamic acid due to inconsistent composition and unsupported health benefits, but DMG itself remained legal (Cupp et al. 2003). Today, DMG is recognized not as a vitamin but as a natural metabolite in one carbon metabolism.
Biosynthesis vs. dietary uptake

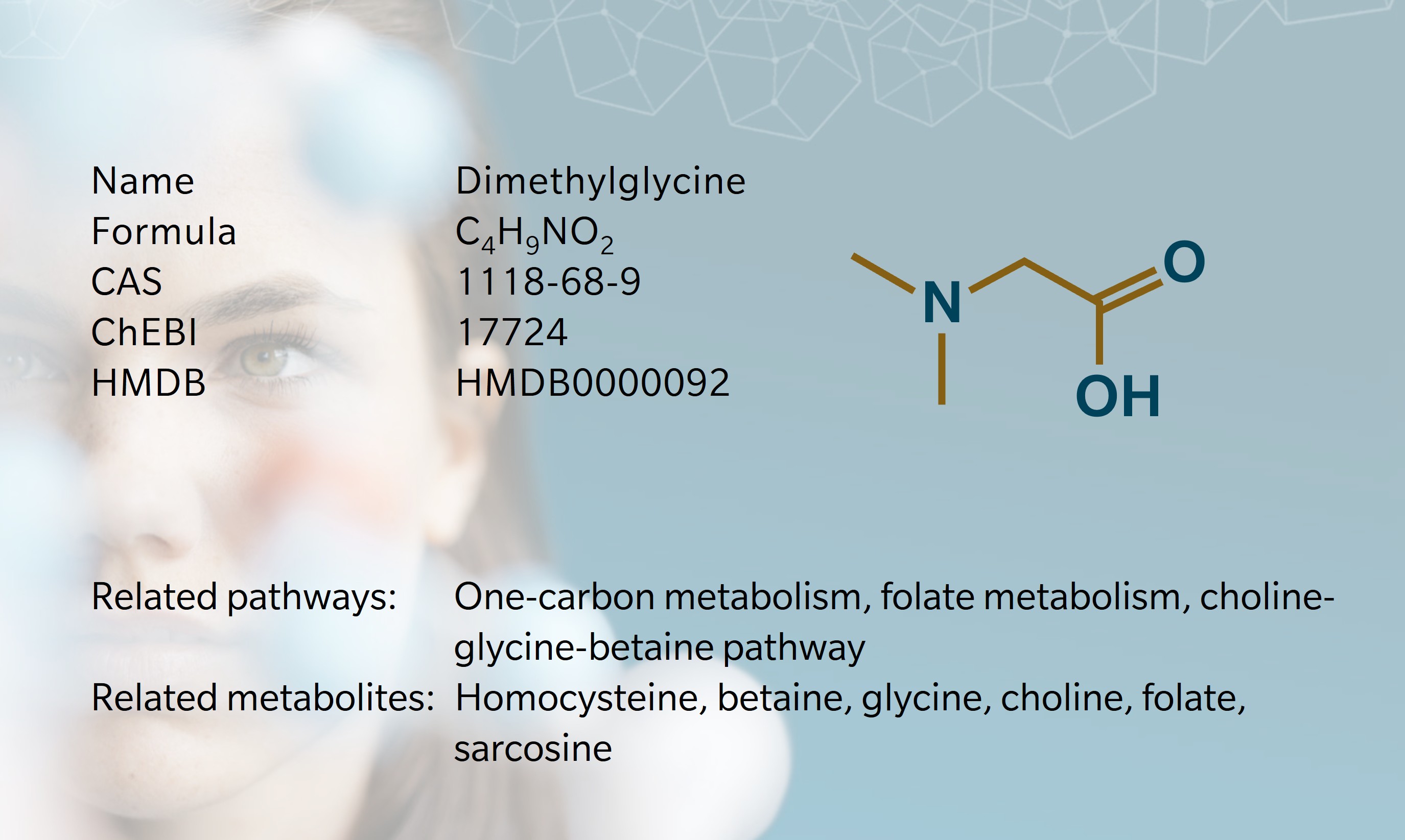
DMG is a naturally occurring amino acid derivative present in the cells of all plants and animals. Small amounts are obtained through the diet, particularly from legumes, grains, and meat (Yao et al. 2022; Cupp et al. 2003). Dietary DMG that is not immediately metabolized in the liver enters the circulation and is delivered to peripheral tissues (Svingen et al. 2013)
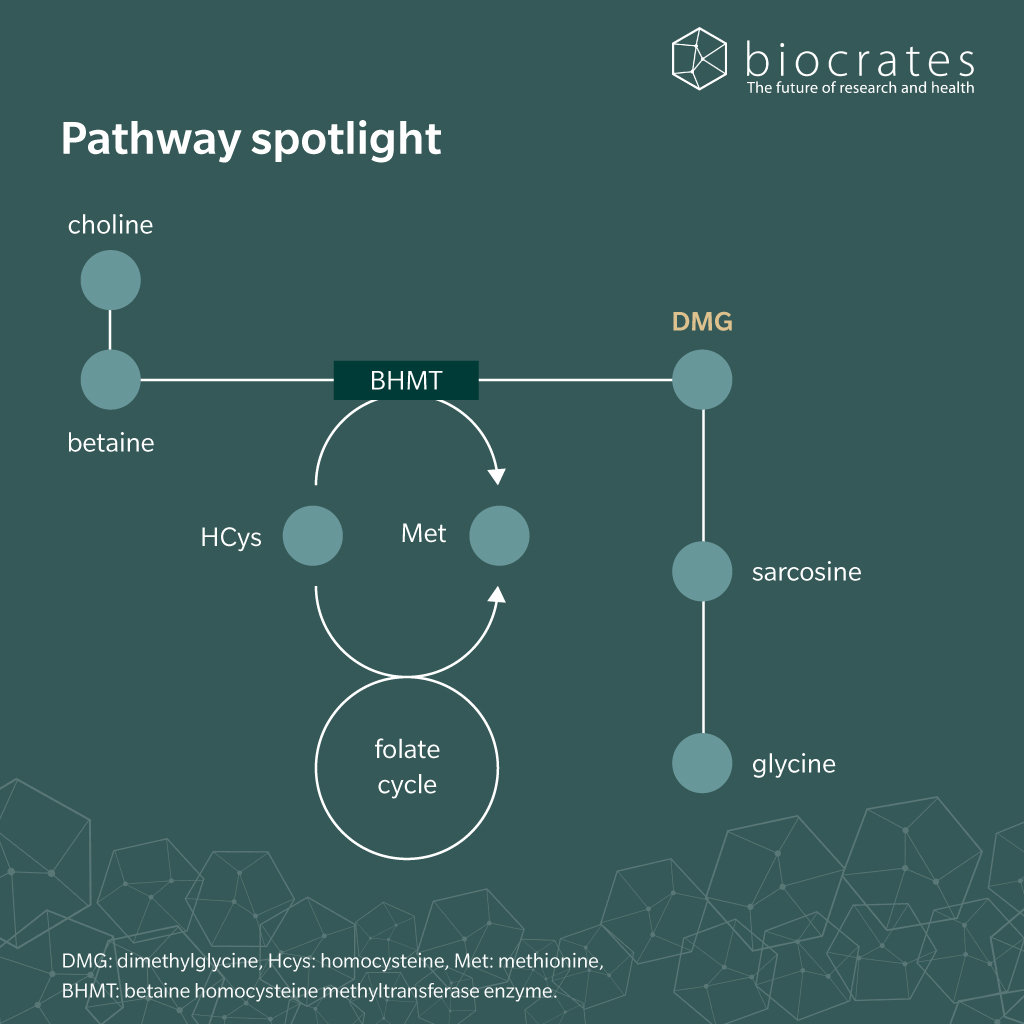
In humans, most DMG arises from endogenous metabolism rather than direct dietary intake (Cupp et al. 2003). DMG is formed when choline is metabolized along the choline–betaine–glycine pathway. A key step in this path is the remethylation of homocysteine mainly in liver and kidney tissue: the enzyme betaine homocysteine methyltransferase (BHMT) transfers a methyl group from betaine to homocysteine, generating methionine and DMG (McGregor et al. 2001). Notably, DMG inhibits BHMT via negative a feedback loop (Svingen et al. 2013).
Once formed, DMG is rapidly oxidized in mitochondria by dimethylglycine dehydrogenase (DMGDH) to produce sarcosine, linking DMG metabolism directly to the folate dependent one carbon cycle (McAndrew et al. 2008). Under normal physiological conditions, circulating DMG levels remain low. A small fraction of unmetabolized DMG is excreted in urine (Svingen et al. 2013). Elevated DMG occurs when DMGDH function is impaired (McAndrew et al. 2008) or when BHMT activity is altered (McGregor et al. 2001).
Dimethylglycine and cardiology
Observational cohort studies consistently link plasma DMG levels with cardiovascular outcomes. In patients with recent acute coronary syndrome, higher baseline DMG was associated with increased risk of acute myocardial infarction, heart failure, and death over ~2.5 years (Lever et al. 2012). Similarly, in a large cohort with stable angina pectoris, DMG correlated with traditional coronary artery disease risk factors and independently predicted incident myocardial infarction during ~4.6 years of follow up (Svingen et al. 2013).
Furthermore, two cohorts of patients with suspected stable angina pectoris or acute myocardial infarction showed that elevated DMG was associated with higher all-cause and cardiovascular mortality, with stronger effects in acute myocardial infarction. DMG improved risk prediction in both groups (Svingen et al. 2015).
In pregnant women carrying fetuses with confirmed congenital heart disease, decreased maternal serum DMG emerged as a potential prenatal diagnostic marker (Xie et al. 2025). Mechanistically, high plasma DMG levels may be linked to the regulation of lipid and energy metabolism.
Experimental evidence suggests that activation of peroxisome proliferator activated receptor alpha (PPARα) reduces transcription of enzymes involved in DMG catabolism, potentially contributing to elevated circulating DMG concentrations (Svingen et al. 2015). However, the exact biological mechanisms underlying the observed associations between DMG and cardiovascular outcomes remain to be fully elucidated.
Dimethylglycine and diabetes
Observational human data indicate that low plasma DMG is associated with higher blood glucose. A genome-wide-association study identified a strong signal at the DMGDH locus that associated with lower DMG and higher insulin, HOMA IR and incident diabetes risk. This suggests that DMG deficiency may contribute to diabetes development and that DMGDH inhibition or DMG supplementation could be explored therapeutically (Magnusson et al. 2015). Additional metabolomics work shows DMG levels differ between normal glucose tolerance and impaired glucose regulation, positioning DMG as a risk associated marker in early dysglycemia (Liu et al. 2024).
Beyond these findings, maternal metabolic studies indicate that mothers carrying twins with low plasma DMG/betaine ratios have a decreased risk of gestational diabetes, suggesting that DMG-betaine balance may modulate glucose metabolism during pregnancy (Gong et al. 2021). Although all human evidence is observational and mechanistic or interventional studies are missing. The metabolic link becomes even clearer in the context of liver disease. The same ratio, this time assessed directly in hepatic tissue, proved highly relevant in a study of metabolic dysfunction associated steatotic liver disease (MASLD) in mice. The impaired DMG/betaine ratio was closely linked to disrupted one carbon remethylation (Pacana et al. 2015). These liver findings support the idea that altered DMG metabolism is part of a broader metabolic dysfunction relevant to both hepatic disease and diabetes.
Dimethylglycine and neurology
DMG supplements are often advertised as supporting cognitive function, mood, memory, or neurotransmitter synthesis, but scientific evidence for neurological benefits remains limited. Preclinical data show some promising mechanistic effects: in a BTBR mouse model of autism spectrum disorder (ASD), supplementation with DMG and B vitamins reduced ASD like behaviors, potentially through multifactor effects including decreased oxidative stress and inflammation, improved gut microbiota and gut permeability, altered body composition, reduced hepatic steatosis, and enhanced mitochondrial function in both liver and brain (Cimmino et al. 2025). However, these findings did not translate into clinical benefit. A double blind, placebo controlled trial in children with autism or pervasive developmental disorder reported no behavioral improvements in 33 of 37 participants receiving DMG (Kern et al. 2001). Similarly, a randomized, placebo controlled pilot study in patients with progressive multiple sclerosis found no effects of DMG on disability, fatigue, or cognitive and motor performance (Wolfsegger et al. 2021). Recent data strengthen the mechanistic rationale for DMG in neurodegeneration, particularly Alzheimer’s disease (AD). DMG was identified as the most effective of twelve metabolites in restoring catalase function, a key antioxidant enzyme impaired in AD, by preventing its aggregation, enhancing activity, stabilizing its native fold, and reducing amyloid like fibrils. Together, these effects suggest that DMG may help counter oxidative stress, a major driver of AD pathology (Devi et al. 2026). However, despite these mechanistic insights, human studies, including interventional trials or metabolomics guided supplementation studies, are still lacking, and no clinical evidence currently supports neurological benefits of DMG.
Dimethylglycine and immunity
DMG shows consistent immunomodulatory effects in animal studies, but human evidence remains limited. In a rabbit model, DMG supplementation markedly increased antibody titers to influenza and Salmonella typhi vaccines and enhanced lymphocyte proliferation, indicating stronger humoral and cellular immunity (Reap et al. 1990). A feline study also reported immunological shifts after DMG-containing supplementation, with reduced neutrophils and increased lymphocytes, possibly reflecting changes in B cell or NK cell activity (Shahril Agus et al. 2025). DMG may further influence immune tolerance: in a rat model of recurrent implantation failure, reduced DMG was linked to lower Treg cell levels and impaired endometrial receptivity, whereas supplementation helped restore immune balance (Liu et al. 2026).
In humans, only one small double blind trial from 1981 showed increased antibody responses and higher leukocyte inhibition factor activity after DMG intake, suggesting enhanced immunity (Graber et al. 1981). However, this finding has never been replicated, and no modern clinical trials confirm immune boosting effects. Thus, while animal data are supportive, evidence in humans remains preliminary and inconclusive.
Dimethylglycine and oncology
Evidence on an influence of DMG in cancer studies is mixed and appears highly context dependent. Large cohort studies report no association between circulating DMG levels and cancer risk, including two nested case-control studies on pancreatic cancer in Asian populations (Huang et al. 2017) and colorectal cancer in the European Prospective Investigation into Cancer and Nutrition (EPIC) cohort (Nitter et al. 2014). In contrast, DMG is elevated in several tumor related settings. Urinary DMG was significantly increased in hepatocellular carcinoma (HCC) in a West African cohort and correlated with clinical stage, suggesting potential utility for surveillance (Ladep et al. 2014). Higher DMG has also been detected in esophageal tumor margins (Jianyong et al. 2017) and in fecal samples of colorectal cancer patients (Lin et al. 2016). One population study in individuals with hypertension reported a positive association between serum DMG and overall cancer risk (Zhang et al. 2023).
These divergent findings may reflect differences in population characteristics, disease stage, or sample types. Mechanistically, DMG participates in one carbon metabolism, supporting amino acid and nucleotide synthesis and cellular methylation reactions. When primary dietary methyl donors (choline, methionine, creatine) are insufficient, DMG can act as a secondary methyl donor. Because dysregulated methylation is a hallmark of cancer, shifts in DMG availability may influence tumor biology, although causal evidence is still limited (Zhang et al. 2023).
Dimethylglycine and 5P medicine
DMG is a molecular entry point into individualized, systems-oriented healthcare aligning with the principles of 5P medicine. Altered DMG levels have been linked to cardiometabolic risk and conditions such as myocardial infarction, diabetes (Magnusson et al. 2015), and metabolic dysfunction-associated liver disease (Pacana et al. 2015). Hence, DMG is positioned as potential early predictive marker even though causal evidence is still missing. Disease specific and matrix dependent patterns, like elevated urinary DMG in hepatocellular carcinoma (Ladep et al. 2014), increased fecal DMG in colorectal cancer (Lin et al. 2016), or reduced maternal DMG in pregnancies with congenital heart disease (Xie et al. 2025), highlight its relevance for more precise diagnostics or stratification. Because DMG metabolism is shaped by nutrition (Yao et al. 2022; Cupp et al. 2003), liver and kidney function (McGregor et al. 2001), genetics (McAndrew et al. 2008), and microbiome (Cimmino et al. 2025; Liu et al. 2024; Wang et al. 2022), individual interpretation is essential. Overall, DMG reflects interconnected pathways such as methylation, mitochondrial function, and cardiometabolic regulation, making it a valuable indicator of metabolic stress and early dysregulation.
References
Cimmino, F. et al.: Autism spectrum disorders and nutritional interventions: dimethylglycine and B-vitamins effects on behaviour, inflammation, microbiota and mitochondria in liver and brain synapses (2025) Biomedicine & pharmacotherapy | https://doi.org/10.1016/j.biopha.2025.118477.
Cupp, M.J. et al.: Dimethylglycine (N,N-Dimethylglycine) (2003) | https://doi.org/10.1007/978-1-59259-303-3_9.
Devi, A.P. et al.: Dimethylglycine as a Potent Modulator of Catalase Stability and Activity in Alzheimer’s Disease (2026) Biophysica | https://doi.org/10.3390/biophysica6010002.
Gong, X. et al.: Maternal Plasma Betaine in Middle Pregnancy Was Associated with Decreased Risk of GDM in Twin Pregnancy: A Cohort Study (2021) Diabetes, metabolic syndrome and obesity : targets and therapy | https://doi.org/10.2147/DMSO.S312334.
Graber, C.D. et al.: Immunomodulating properties of dimethylglycine in humans (1981) The Journal of Infectious Diseases | https://doi.org/10.1093/infdis/143.1.101.
Huang, J. et al.: Abstract 2273: Serum choline, methionine, betaine, dimethylglycine, and trimethylamine-N-oxide in relation to pancreatic cancer risk in two nested case-control studies in Asian populations (2017) Cancer Research | https://doi.org/10.1158/1538-7445.AM2017-2273.
Jianyong, Z. et al.: Rapid discrimination of human oesophageal squamous cell carcinoma by mass spectrometry based on differences in amino acid metabolism (2017) Scientific Reports | https://doi.org/10.1038/s41598-017-03375-8.
Kern, J.K. et al.: Effectiveness of N,N-dimethylglycine in autism and pervasive developmental disorder (2001) Journal of child neurology | https://doi.org/10.1177/088307380101600303.
Ladep, N.G. et al.: Discovery and validation of urinary metabotypes for the diagnosis of hepatocellular carcinoma in West Africans (2014) Hepatology | https://doi.org/10.1002/hep.27264.
Lever, M. et al.: Betaine and secondary events in an acute coronary syndrome cohort (2012) PLOS ONE | https://doi.org/10.1371/journal.pone.0037883.
Lin, Y. et al.: NMR-based fecal metabolomics fingerprinting as predictors of earlier diagnosis in patients with colorectal cancer (2016) Oncotarget | https://doi.org/10.18632/oncotarget.8762.
Liu, F.-T. et al.: Endometrial microbiota-dimethylglycine-Treg cell axis affects endometrial receptivity in recurrent implantation failure (2026) Science China. Life sciences | https://doi.org/10.1007/s11427-025-3138-x.
Liu, Y. et al.: Changes in Isoleucine, Sarcosine, and Dimethylglycine During OGTT as Risk Factors for Diabetes (2024) The Journal of Clinical Endocrinology & Metabolism | https://doi.org/10.1210/clinem/dgae018.
Magnusson, M. et al.: Dimethylglycine Deficiency and the Development of Diabetes (2015) Diabetes | https://doi.org/10.2337/db14-1863.
McAndrew, R.P. et al.: Molecular basis of dimethylglycine dehydrogenase deficiency associated with pathogenic variant H109R (2008) Journal of inherited metabolic disease | https://doi.org/10.1007/s10545-008-0999-2.
McGregor, D.O. et al.: Dimethylglycine accumulates in uremia and predicts elevated plasma homocysteine concentrations (2001) Kidney International | https://doi.org/10.1046/j.1523-1755.2001.00743.x.
Nitter, M. et al.: Plasma methionine, choline, betaine, and dimethylglycine in relation to colorectal cancer risk in the European Prospective Investigation into Cancer and Nutrition (EPIC) (2014) Annals of oncology : official journal of the European Society for Medical Oncology | https://doi.org/10.1093/annonc/mdu185.
Pacana, T. et al.: Dysregulated Hepatic Methionine Metabolism Drives Homocysteine Elevation in Diet-Induced Nonalcoholic Fatty Liver Disease (2015) PLOS ONE | https://doi.org/10.1371/journal.pone.0136822.
Reap, E.A. et al.: Stimulation of the immune response by dimethylglycine, a nontoxic metabolite (1990) The Journal of laboratory and clinical medicine | PMID: 1691258.
Shahril Agus, S.A. et al.: Effect of N, N-Dimethylglycine (DMG) Supplementation on Haematological Parameters and Frequency of CD4+ and CD8+ T Cells in Cats Post-vaccination (2025) Pertanika Journal of Tropical Agricultural Science | https://doi.org/10.47836/pjtas.48.3.12.
Svingen, G.F.T. et al.: Plasma dimethylglycine and risk of incident acute myocardial infarction in patients with stable angina pectoris (2013) Arteriosclerosis, thrombosis, and vascular biology | https://doi.org/10.1161/ATVBAHA.113.301714.
Svingen, G.F.T. et al.: Elevated plasma dimethylglycine is a risk marker of mortality in patients with coronary heart disease (2015) European Journal of Preventive Cardiology | https://doi.org/10.1177/2047487314529351.
Wang, Z. et al.: Heat stress-induced intestinal barrier damage and dimethylglycine alleviates via improving the metabolism function of microbiota gut brain axis (2022) Ecotoxicology and Environmental Safety | https://doi.org/10.1016/j.ecoenv.2022.114053.
Wolfsegger, T. et al.: N,N-Dimethylglycine in patients with progressive multiple sclerosis: result of a pilot double-blind, placebo, controlled randomized clinical trial (2021) Neurological research and practice | https://doi.org/10.1186/s42466-021-00126-z.
Xie, B. et al.: Identification of serum N,N-dimethylglycine as a potential biomarker for prenatal diagnosis of congenital heart disease using 1HNMR and UPLC-MS/MS metabonomics (2025) Analytical and Bioanalytical Chemistry | https://doi.org/10.1007/s00216-025-06084-8.
Yao, H. et al.: Effects of dietary dimethylglycine supplementation on laying performance, egg quality, and tissue index of hens during late laying period (2022) Poultry Science | https://doi.org/10.1016/j.psj.2021.101610.
Zhang, H. et al.: The association of serum dimethylglycine with the risk of cancer (2023) | https://doi.org/10.21203/rs.3.rs-3371052/v1.