- History & Evolution
- Biosynthesis & dietary uptake
- EPA and the microbiome
- EPA and inflammation
- EPA and neurology
- EPA and oncology
- EPA and 5P medicine
- References
History & Evolution
1957: first isolated (Klenk, E. and Eberhagen, D., 1957) | 1964: EPA linked to prostaglandin production (Bergström, S. et al., 1964) | 1978: discovery of cardiovascular protective effects
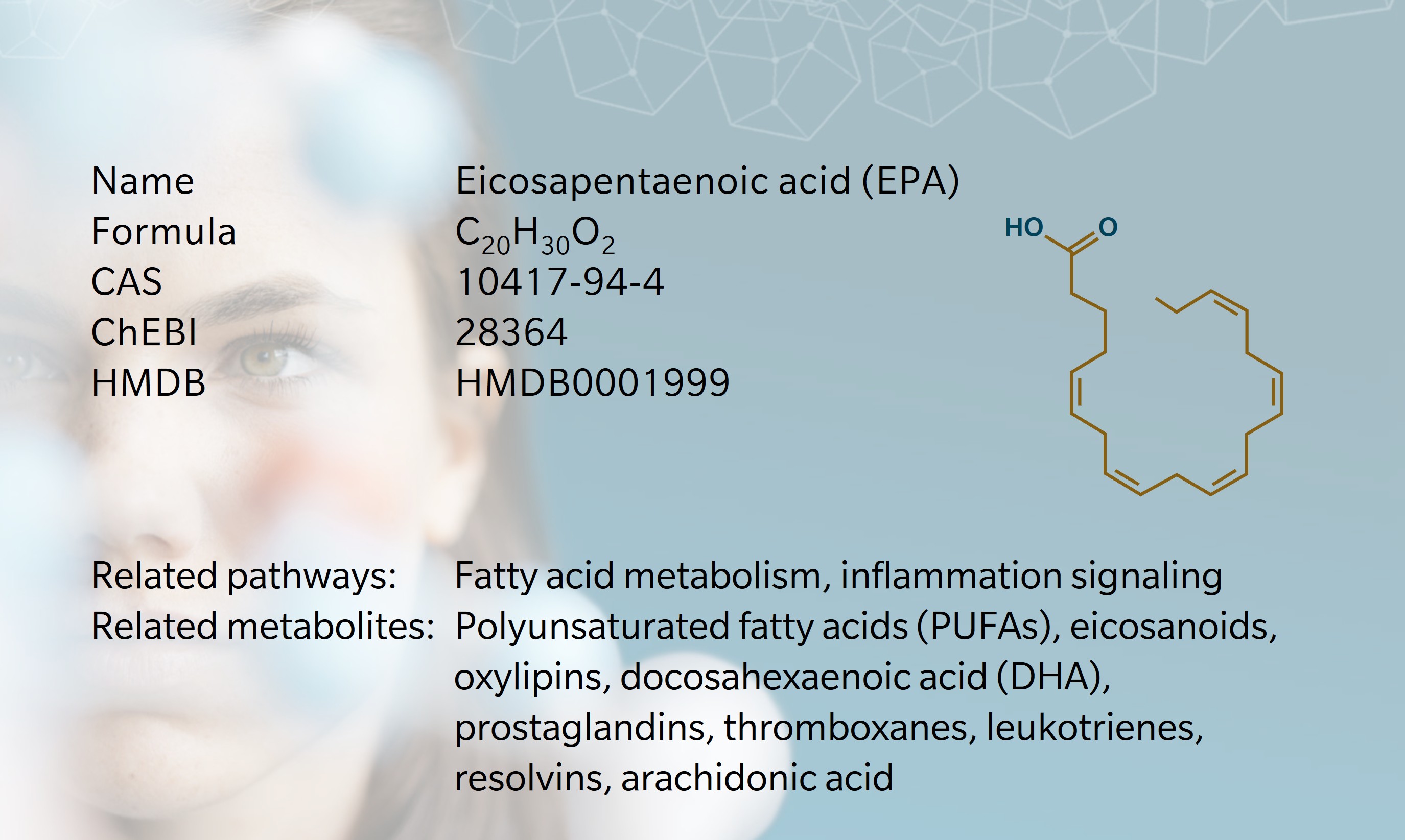
Eicosapentaenoic acid (EPA) is a long-chain omega-3 polyunsaturated fatty acid (PUFA) and a precursor to many lipid signaling molecules involved in inflammation and cardiometabolic regulation. The name reflects its structure: “eicosa” refers to its 20 carbon atoms, while “pentaenoic” refers to five double bonds (Castro, L. et al., 2016).
EPA was first isolated from cod liver oil in 1957 by Klenk and Eberhagen (Klenk, E. and Eberhagen, D., 1957). At the time, lipid research focused on omega-6 fatty acids, particularly arachidonic acid (AA) and linoleic acid, as their roles in cholesterol metabolism and prostaglandin synthesis made them more compelling targets (Spector, A. and Kim, H., 2019). Although Bergström later showed that EPA could also be enzymatically converted into prostaglandin E3 (Bergström, S. et al., 1964), this pathway attracted little attention and interesting early findings on omega-3 fatty acids were largely overlooked.
This changed in 1978, when Dyerberg and Bang observed notably low rates of myocardial infarction among Greenland Eskimos, who typically followed a diet of marine lipids rich in EPA and docosahexaenoic acid (DHA) (Dyerberg, J. and Bang, O., 1978).
By the 1980s, epidemiological and clinical studies linked diets rich in marine omega-3 fatty acids to lower rates of cardiovascular disease, putting EPA at the heart of vascular biology. EPA has since been shown to have protective effects against wide-ranging diseases, including obesity, diabetes, chronic obstructive pulmonary disease and certain cancers (Watabe, S. et al., 2024).
EPA is found primarily in marine microalgae, fish and other marine animals, where it accumulates through the food chain.
Biosynthesis vs. dietary uptake
In humans, EPA comes mainly from the diet, particularly oily fish, seafood and fish-based supplements. However, this is not a reliable source. Fish do not produce EPA themselves but obtain it from algae, so their EPA content varies depending on their diet and environment (Watabe, S. et al., 2024). Concerns about sustainability and contamination of fish and fish-derived supplements have increased interest in alternative sources such as algal oils (Sousa, S. et al., 2024).
EPA can also be produced endogenously, but only in small amounts. It is synthesized from the essential fatty acid alpha-linolenic acid (ALA), found in foods such as flaxseed, walnuts and vegetable oils (Watabe, S. et al., 2024). This conversion takes place mainly in the liver through a series of enzymatic reactions. The process is quite inefficient: only 1-15% of ALA is converted to EPA, and this can be further reduced by alcohol consumption, smoking, inactivity and dietary factors (Sousa, S. et al., 2024).
As a result, endogenous synthesis does not make a significant contribution to circulating EPA and, for most people, dietary intake is the main source. Recommended daily intake is around 250-500 mg per day (together with downstream metabolite DHA), rising to 700-1000 mg for pregnant and lactating women (Sousa, S. et al., 2024).
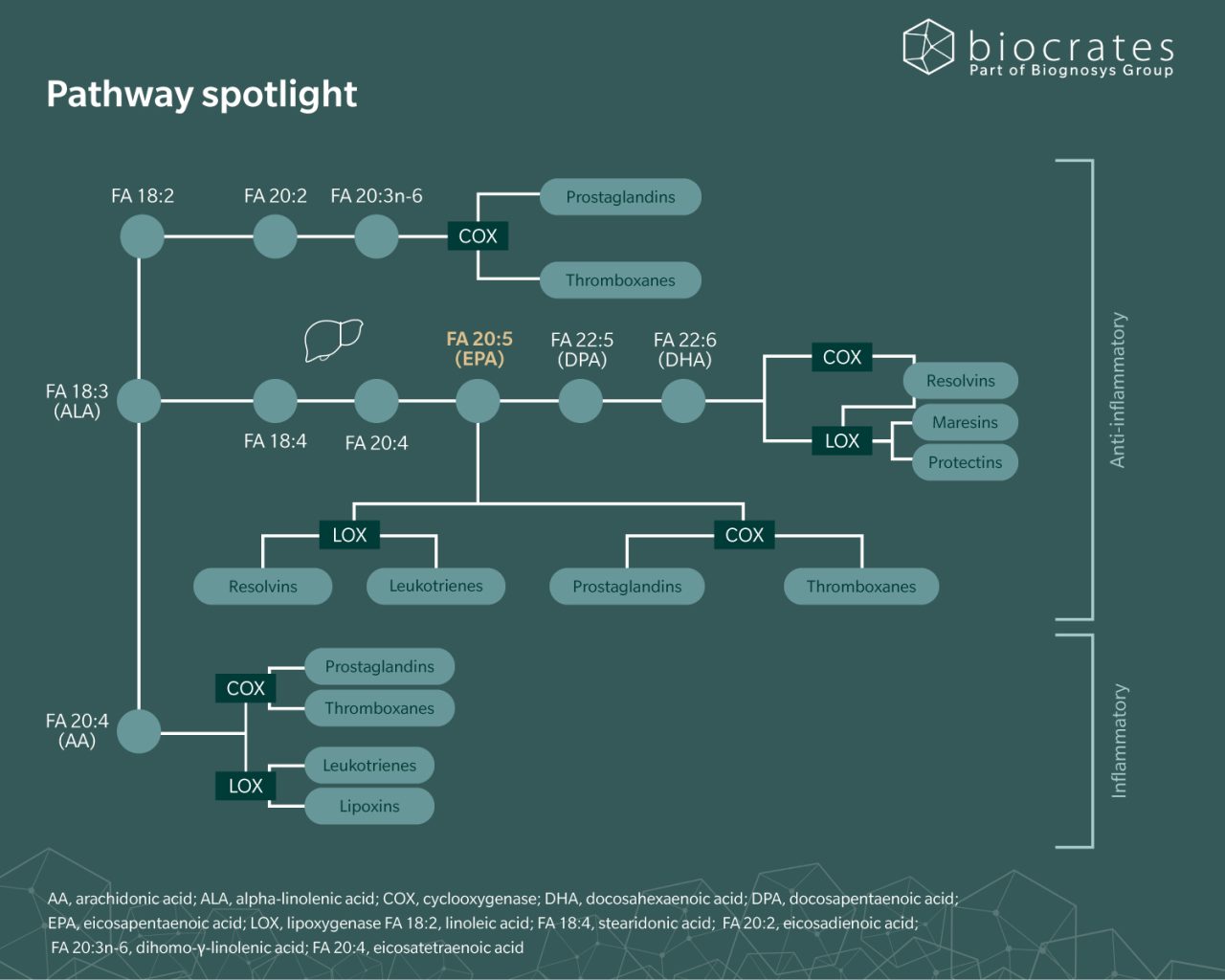
Once in circulation, EPA is incorporated into cell membranes where it can be converted into lipid mediators, including prostaglandins, leukotrienes and resolvins (Igarashi, M. et al., 2013).
EPA and the microbiome
Research shows that EPA interacts with the gut microbiome, directly affecting bacterial growth, diversity and survival. As with other PUFAs, EPA has been shown in experimental settings to have an antibacterial effect against Gram-positive species such as Bacillus cereus and Staphylococcus aureus, which are both causes of infection (Le, PNT. and Desbois, A., 2017).
Studies report a decrease in the Firmicutes/Bacteroidetes ratio and increases in genera such as Bifidobacterium, Lachnospira, Roseburia and Lactobacillus (Amedei, A. et al., 2025). EPA has also been found to modulate levels of pro-inflammatory molecules and affect short-chain fatty acid (SCFA) concentrations (Fu, Y. et al., 2021). These effects are associated with improved cardiovascular risk profiles.
In turn, beneficial bacteria including Bifidobacterium, Lactobacillus and Akkermansia have been shown to influence EPA absorption and metabolism, indicating a two-way interaction between EPA and the gut microbiome (Amedei, A. et al., 2025).
EPA and inflammation
Inflammation is one of the main pathways through which EPA affects human health.
As a precursor to lipid signaling molecules, EPA influences how inflammatory mediators are produced. In cell membranes, it partly replaces omega-6 fatty acids such as arachidonic acid, changing the pool of fatty acids available for enzymatic conversion and leading to the production of eicosanoids that are generally less pro-inflammatory (Crupi, R. and Cuzzocrea, S., 2022). EPA is also converted into specialized pro-resolving mediators, such as resolvins, which help limit and resolve inflammation. More broadly, these changes in membrane composition influence cell signaling, gene expression and the production of cytokines, adhesion molecules and other inflammatory mediators (Calder, P., 2010).
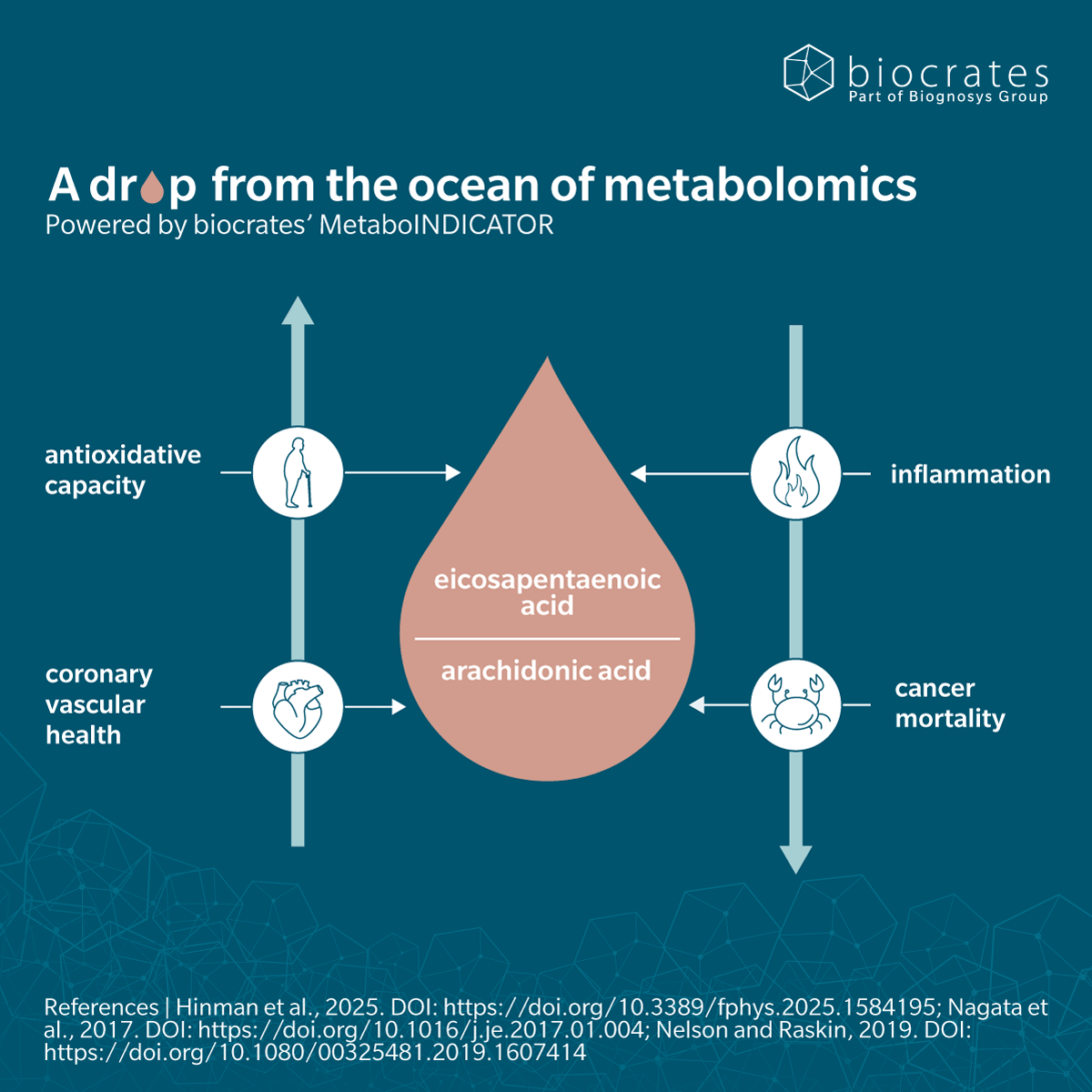
These pathways are also influenced by the exposome. Environmental factors such as diet, pollutants and oxidative stress can alter fatty acid composition and downstream inflammatory signaling (Hinman, J. et al., 2025). Studies show that higher omega-3 status, including EPA levels, is associated with increased pro-resolving and reduced pro-inflammatory lipid mediator responses following exposures such as air pollution, highlighting EPA’s role in shaping inflammatory responses to environmental stressors (Chen, H. et al., 2025).
Clinically, this translates into measurable effects on disease-related inflammation. EPA has been shown to reduce pro-inflammatory cytokines and oxidative stress, improve endothelial function and support stabilization of atherosclerotic plaques, all of which are relevant to cardiovascular risk (Crupi, R. and Cuzzocrea, S., 2022). In inflammatory conditions such as rheumatoid arthritis, higher intakes of EPA (typically with DHA) are associated with reduced inflammatory activity, although relatively high and sustained doses are often required (Calder, P., 2010). A 2024 literature review found that EPA is linked to improved metabolic and inflammatory profiles in non-communicable diet-related conditions such as obesity, diabetes and cardiovascular disease (Banaszak, M. et al., 2024).
A 2021 study using targeted lipidomics linked EPA’s anti-inflammatory effects to improvements in depression (Borsini, A. et al., 2021). Using a “depression in a dish” model of human hippocampal cells, researchers showed that EPA-derived lipid mediators protected neurons from inflammation-related damage and supported neurogenesis. In patients with major depressive disorder, higher levels of these metabolites were associated with reduced symptom severity, suggesting a mechanistic link between EPA, inflammation and depression.
EPA and neurology
EPA has been studied in neurology because of its effect on synaptic function, neuroinflammation and neuronal survival. In a preclinical study, EPA was taken up by both neurons and glial cells (Kawashima, A. et al., 2010). This increased long-term potentiation in the hippocampus and activated intracellular signaling involving phosphoinositide 3-kinase (PI3K) and protein kinase B (Akt), while reducing markers of cell death. These findings suggest a potential role in protecting against neurodegeneration and support interest in EPA in conditions such as Alzheimer’s disease, Huntington’s disease and schizophrenia.
More recent work has looked at EPA in neurodevelopmental disorders and epilepsy, where inflammation, oxidative stress and synaptic dysfunction are recurring factors (Li, M. et al., 2025). Omega-3 fatty acids, including EPA, may help in attention deficit hyperactivity disorder (ADHD), autism spectrum disorder and Tourette syndrome, but the evidence is mixed and clinical effects are often modest. For example, in ADHD, some studies report improvement in attention or behavior from higher EPA doses, while others find no clear benefit. In epilepsy, EPA and other omega-3 fatty acids have been explored mainly as adjuncts rather than stand-alone treatments, with some studies reporting reduced inflammatory markers or seizure frequency.
Omics studies suggest that lipid dysregulation, including EPA, may contribute to neurological disease progression. For example, in a 2025 longitudinal metabolomic-lipidomic study of 767 people with multiple sclerosis, decreased serum EPA was associated with slower walking speed and reduced manual dexterity (Noroozi, R. et al., 2026).
A 2026 study found that after repetitive mild traumatic brain injury, EPA accumulates in the brain under baseline conditions but is then depleted during injury-related metabolic stress (Karakaya, E. et al., 2026). This was associated with impaired endothelial repair, reduced angiogenic signaling and vascular and cognitive decline.
These findings make EPA a metabolite of interest as a marker of and potential therapeutic target in neuroinflammatory and neurovascular processes.
EPA and oncology
The relationship between EPA and cancer is unclear. The metabolite has been shown to suppress proliferation in colon, pancreatic, breast, esophageal and other cancer cell lines (Mizoguchi, K. et al., 2014), and EPA-derived resolvins seem to have an anti-tumor effect (Kiyasu, Y. et al., 2024).
However, clinical studies show mixed results. In animal models, for example, fish oil has been shown to both suppress colorectal carcinogenesis and to promote colitis-associated cancer (Kiyasu, Y. et al., 2024).
Evidence in patients is also limited. Small studies in patients undergoing chemotherapy report changes in inflammatory markers and immune function with EPA (often combined with DHA) (Munhoz, J. et al., 2025). However, findings are inconsistent, and there is currently insufficient evidence to support routine use in cancer care.
EPA and 5P medicine
EPA’s links to diet, genetics and inflammation make it an interesting metabolite for the study of chronic, non-communicable diseases, which are a top priority in population health. In epidemiological research, circulating omega-3 fatty acids, including EPA, are used as objective biomarkers of dietary intake and have been associated with cardiovascular outcomes and mortality risk across large cohorts and meta-analyses (Jiang, H. et al., 2022).
Studies also show that EPA’s effects depend on both exposure (diet) and host biology (genetics), with implications for precision medicine. For example, in the seAFOod polyp prevention trial, EPA supplementation was found to reduce adenoma risk by around 50% in individuals with a specific FADS variant, while those without the allele saw no benefit (Sun, G. et al., 2024).
EPA also seems to play a role in aging, making it increasingly relevant as populations age and the burden of chronic disease grows (Hinman, J. et al., 2025). Studies show changes in PUFA profiles and inflammatory and oxidative stress markers across the lifespan (Aiello, A. et al., 2024). In long-lived women, a lower AA to EPA ratio was associated with higher antioxidant capacity (Hinman, J. et al., 2025).
References
Aiello, A. et al. (2024). Polyunsaturated fatty acid status and markers of oxidative stress and inflammation across the lifespan: A cross-sectional study in a cohort with long-lived individuals. Experimental Gerontology, 195, 112531. DOI: https://doi.org/10.1016/j.exger.2024.112531.
Amedei, A. et al. (2025). Potential and Future Therapeutic Applications of Eicosapentaenoic/Docosahexaenoic Acid and Probiotics in Chronic Low-Grade Inflammation. Biomedicines, 13(10), 2428. DOI: https://doi.org/10.3390/biomedicines13102428.
Banaszak, M. et al. (2024). Role of Omega-3 fatty acids eicosapentaenoic (EPA) and docosahexaenoic (DHA) as modulatory and anti-inflammatory agents in noncommunicable diet-related diseases – Reports from the last 10 years. Clinical Nutrition ESPEN, 63, 240-258. DOI: https://doi.org/10.1016/j.clnesp.2024.06.053.
Bergström, S. et al. (1964). The Enzymatic Conversion of Essential Fatty Acids into Prostaglandins: PROSTAGLANDINS AND RELATED FACTORS 34. Journal of Biological Chemistry, 239, PC4006-PC4008. DOI: https://doi.org/10.1016/S0021-9258(18)91234-2.
Borsini, A. et al. (2021). Omega-3 polyunsaturated fatty acids protect against inflammation through production of LOX and CYP450 lipid mediators: relevance for major depression and for human hippocampal neurogenesis. Molecular Psychiatry, 26, 6773–6788. DOI: https://doi.org/10.1038/s41380-021-01160-8.
Calder, P. (2010). Omega-3 Fatty Acids and Inflammatory Processes. Nutrients, 2(3), 355–374. DOI: https://doi.org/10.3390/nu2030355.
Castro, L. et al. (2016). Long-chain polyunsaturated fatty acid biosynthesis in chordates: Insights into the evolution of Fads and Elovl gene repertoire. Progress in Lipid Research, 62, 25-40. DOI: https://doi.org/10.1016/j.plipres.2016.01.001.
Chen, H. et al. (2025). Omega-3 Fatty Acid Intake and Oxylipin Production in Response to Short-Term Ambient Air Pollution Exposure in Healthy Adults. Toxics, 13(12), 1063. DOI: https://doi.org/10.3390/toxics13121063.
Crupi, R. and Cuzzocrea, S. (2022). Role of EPA in Inflammation: Mechanisms, Effects, and Clinical Relevance. Biomolecules, 12(2), 242. DOI: https://doi.org/10.3390/biom12020242.
Dyerberg, J. and Bang, O. (1978). Dietary fat and thrombosis. The Lancet, 311(8056), 152. DOI: https://doi.org/10.1016/S0140-6736(78)90448-8.
Fu, Y. et al. (2021). Associations among Dietary Omega-3 Polyunsaturated Fatty Acids, the Gut Microbiota, and Intestinal Immunity. Mediators Inflamm, 8879227. DOI: https://doi.org/10.1155/2021/8879227.
Hinman, J. et al. (2025). Effects of molecular interactions between the exposome and oxylipin metabolism on healthspan. Front. Physiol., 16, DOI: https://doi.org/10.3389/fphys.2025.1584195.
Igarashi, M. et al. (2013). Kinetics of eicosapentaenoic acid in brain, heart and liver of conscious rats fed a high n-3 PUFA containing diet. Prostaglandins Leukot Essent Fatty Acids, 89(6), 403–412. DOI: https://doi.org/10.1016/j.plefa.2013.09.004.
Jiang, H. et al. (2022). Omega-3 polyunsaturated fatty acid biomarkers and risk of type 2 diabetes, cardiovascular disease, cancer, and mortality. Clin Nutr, 41(8), 798-1807. DOI: https://doi.org/10.1016/j.clnu.2022.06.034.
Karakaya, E. et al. (2026). Eicosapentaenoic acid reprograms cerebrovascular metabolism and impairs repair after brain injury, with relevance to chronic traumatic encephalopathy. Cell Reports, 117135. DOI: https://doi.org/10.1016/j.celrep.2026.117135.
Kawashima, A. et al. (2010). Effects of eicosapentaenoic acid on synaptic plasticity, fatty acid profile and phosphoinositide 3-kinase signaling in rat hippocampus and differentiated PC12 cells. J Nutr Biochem, 21(4), 268-77. DOI: https://doi.org/10.1016/j.jnutbio.2008.12.015. Epub 2009.
Kiyasu, Y. et al. (2024). EPA, DHA, and resolvin effects on cancer risk: the underexplored mechanisms. Prostaglandins Other Lipid Mediat, 174, 106854. DOI: https://doi.org/10.1016/j.prostaglandins.2024.106854.
Klenk, E. and Eberhagen, D. (1957). [Occurrence of delta5, 8, 11, 14, 17-n-eicosapentanoic acid in cod liver oil and its isolation] (Article in German). Hoppe Seylers Z Physiol Chem, 307(1), 42-8.
Le, PNT. and Desbois, A. (2017). Antibacterial Effect of Eicosapentaenoic Acid against Bacillus cereus and Staphylococcus aureus: Killing Kinetics, Selection for Resistance, and Potential Cellular Target. Mar Drugs, 15(11), 334. DOI: https://doi.org/10.3390/md15110334.
Li, M. et al. (2025). Omega-3 fatty acids: multi-target mechanisms and therapeutic applications in neurodevelopmental disorders and epilepsy. Front. Nutr. , 12, 1598588. DOI: https://doi.org/10.3389/fnut.2025.1598588.
Mizoguchi, K. et al. (2014). Induction of Apoptosis by Eicosapentaenoic Acid in Esophageal Squamous Cell Carcinoma. Anticancer Research December, 34(12), 7145-7149. PMID: 25503142.
Munhoz, J. et al. (2025). Perspective: Implications of Docosahexaenoic Acid and Eicosapentaenoic Acid Supplementation on the Immune System during Cancer Chemotherapy: Perspectives from Current Clinical Evidence. Advances in Nutrition, 16(8), 100464. DOI: https://doi.org/10.1016/j.advnut.2025.100464.
Noroozi, R. et al. (2026). Serum metabolomic-lipidomic signatures track long-term neurological performance in multiple sclerosis. Mol. Neurodegener. Adv., 2(1), DOI: https://doi.org/10.1186/s44477-025-00008-7.
Sousa, S. et al. (2024). Factors impacting the microbial production of eicosapentaenoic acid. Appl Microbiol Biotechnol, 108(1), 368. DOI: https://doi.org/10.1007/s00253-024-13209-z.
Spector, A. and Kim, H. (2019). Emergence of omega-3 fatty acids in biomedical research. Prostaglandins, Leukotrienes and Essential Fatty Acids, 140, 47-50. DOI: https://doi.org/10.1016/j.plefa.2018.11.017.
Sun, G. et al. (2024). Fatty acid desaturase insertion-deletion polymorphism rs66698963 predicts colorectal polyp prevention by the n-3 fatty acid eicosapentaenoic acid: a secondary analysis of the seAFOod polyp prevention trial. Am J Clin Nutr, 120(2), 360-368. DOI: https://doi.org/10.1016/j.ajcnut.2024.06.004.
Watabe, S. et al. (2024). Daily Consumption of α-Linolenic Acid Increases Conversion Efficiency to Eicosapentaenoic Acid in Mice. Nutrients, 16(10), 407. DOI: https://doi.org/10.3390/nu16101407.