Poster gallery
Learn more about biocrates kits, technology and science by gaining access to our posters

Gender differences in precision medicine
Presented at Wellcome conference on Multiomics in Metabolic Disease 2025
Precision medicine builds on differences in genetics, environment, and lifestyle – factors that are most directly captured by metabolomics. Yet stratifying omics data by gender, the most basic level of precision medicine, remains severely underestimated and underutilized.
Our results show that gender-based stratification must be integrated into study design, data processing, and interpretation. Establishing healthy reference ranges is essential for applying metabolomics-powered precision medicine to disease risk, progression, and treatment response in both clinical practice and drug development.

Microbiome research with MxP Quant 1000
Presented at Metabolomics 2025
Microbiome–host interactions are reflected by circulating metabolites in plasma and excreted metabolites in feces and urine. The MxP Quant 1000 provides a standardized, quantitative approach that combines the robustness and sensitivity of targeted solutions with the broad coverage typically associated with untargeted metabolomics.
Combining feces, plasma, and urine enables metabolic pathways to be traced across matrices, providing a more comprehensive view of microbiome–host interactions.

MxP Quant 1000 across biological matrices
Presented at Metabolomics 2025
The detectability of the 1,233 metabolites targeted with the MxP Quant 1000 kit was evaluated across nine matrices. The MxP Quant 1000 kit shows broad applicability across species and biological matrices, with high metabolite coverage and robust analytical precision. Its wide-ranging detectability and low analytical variation make it a powerful tool for quantitative metabolomics in translational research, fundamental science, clinical studies, and drug development.
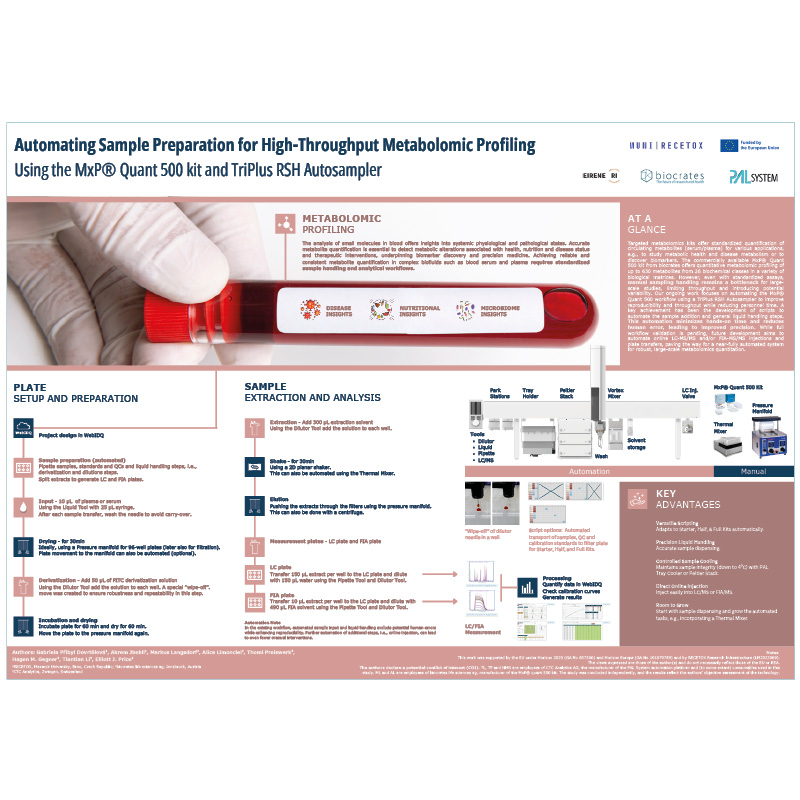
MxP Quant 500 automation on TriPlus RSH Autosampler
Presented at ASMS 2025
Automating sample preparation for metabolomic profiling with the MxP Quant 500 kit and a TriPlus RSH autosampler streamlines complex workflows and reduces manual variability. This setup supports robust, standardized processing of large cohorts while significantly reducing expert hands-on time.
By integrating automation into the MxP Quant 500 workflow, laboratories can achieve higher throughput and precision. This makes large-scale metabolomics more accessible and accelerates biomarker discovery, clinical research, and translational applications.
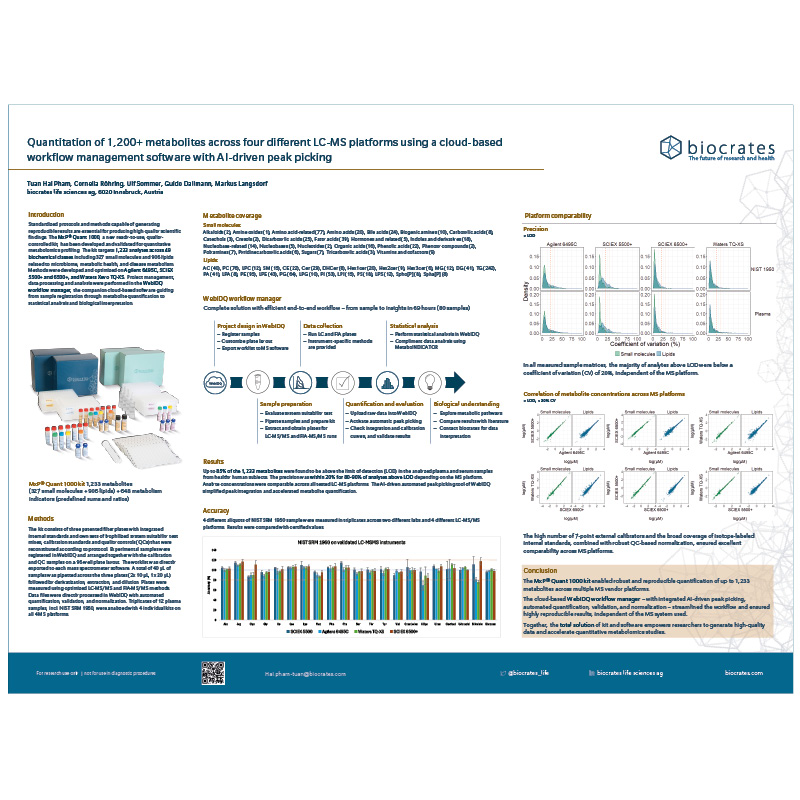
MxP Quant 1000 across MS platforms
Presented at ASMS 2025
The MxP Quant 1000 kit enables robust and reproducible quantification of up to 1,233 metabolites across multiple MS vendor platforms. We compared precision, accuracy, and metabolite concentration correlations across four MS platforms: Agilent 6495C, SCIEX 5500+ and 6500+, and Waters Xevo TQ-XS. Comparable results are guaranteed by the WebIDQ workflow manager, offering standardized cloud-based project management, data processing, and analysis.
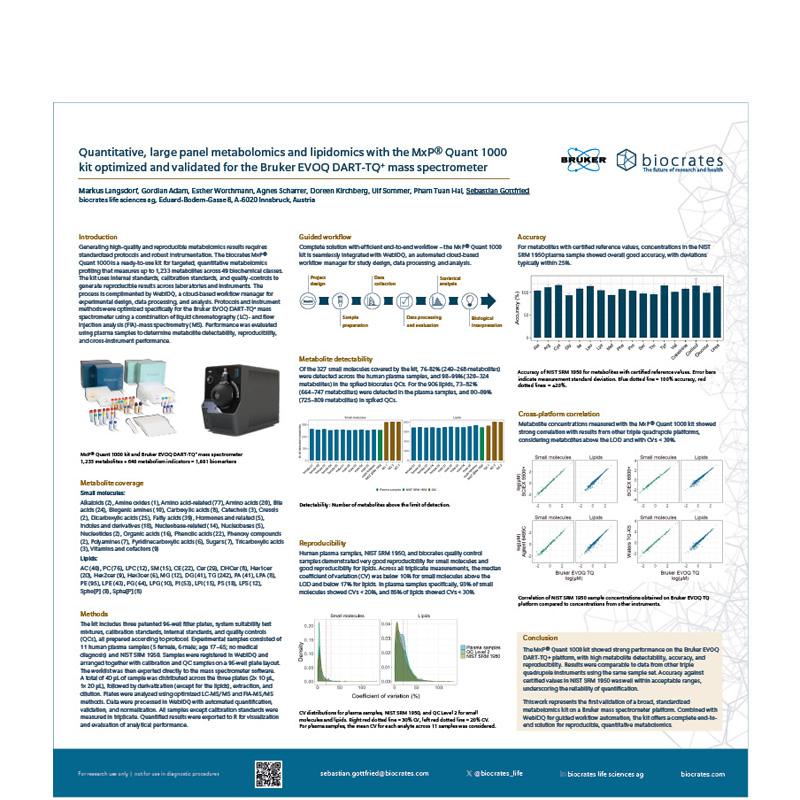
MxP Quant 1000 on EVOQ-DART TQ+
Presented at MSACL 2025
Protocols and instrument methods of the MxP Quant 1000 kit were optimized for the Bruker EVOQ DART-TQ+ mass spectrometer. Performance was evaluated in plasma to assess metabolite detectability, reproducibility, accuracy, and cross-instrument comparability.
The MxP Quant 1000 kit showed strong performance on the Bruker EVOQ DART-TQ+ platform, with high metabolite detectability and reproducibility, comparable to other triple quadrupole systems. Accuracy against NIST SRM 1950 confirmed reliable quantification, marking the first validation of a broad, standardized metabolomics kit on a Bruker MS platform.
